Advertisements
Advertisements
Question
Identify the following method of concentration of ores and explain briefly.
Advertisements
Solution
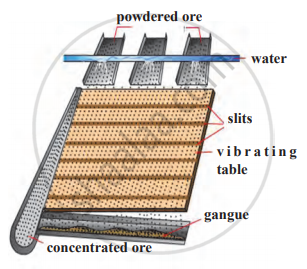
- This method represents Wilfley’s table of concentration of ores.
- Explanation:
- Wilfley’s table is made by fixing narrow, thin and long wooden riffles on an inclined surface.
- The table is given a regular vibrating motion.
- The powdered ore obtained from lumps of the ore using ball mill is fed on the top of Wilfley’s table and running stream of water is also released from the upper side.
- As a result, the lighter gangue particles are carried away by water and heavy ore particles in which proportion of minerals is more, settle between the wooden riffles fixed on the table and get collected on the slits between them.
APPEARS IN
RELATED QUESTIONS
Give the importance of the following for living beings:
Hydrogen
Why are alkali metals kept in kerosene oil?
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their: hydroxide
Define the term ‘metallurgy’. State the processes involved in metallurgy ?
Name the processes involved in refining of ores.
Explain the following terms:
(c) slag
On which factors does the purification of metals depend?
`Cu (OH)2` ................................
................................
`ZnCO_3` ..............................
..............................
Give the ionization reactions of electrolyte used in Hall’s process. write the reaction at the cathode and the anode. Why the anode has to be replaced in this process?
Name a metal which forms a liquid alloy at ordinary temperature.
A compound that is added to lower the fusion temperature of the electrolytic bath in the extraction of aluminium.
What do you observe when hydrogen is passed over heated copper oxide?
In Wilfley table method to separate particles of gangue _______ method is used.
Write the molecular formulae of the following compound.
Stannic oxide
Distinguish between:
Froth floatation - Leaching
Limestone is used as a flux in the extraction of ______.
After partial roasting, the sulphide of copper is reduced by ______.
Calcination is used in metallurgy for the removal of?
