Advertisements
Advertisements
प्रश्न
Identify the following method of concentration of ores and explain briefly.
Advertisements
उत्तर
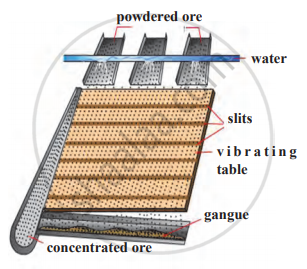
- This method represents Wilfley’s table of concentration of ores.
- Explanation:
- Wilfley’s table is made by fixing narrow, thin and long wooden riffles on an inclined surface.
- The table is given a regular vibrating motion.
- The powdered ore obtained from lumps of the ore using ball mill is fed on the top of Wilfley’s table and running stream of water is also released from the upper side.
- As a result, the lighter gangue particles are carried away by water and heavy ore particles in which proportion of minerals is more, settle between the wooden riffles fixed on the table and get collected on the slits between them.
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Nitrogen
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their: hydroxide
Name:
(a) the processes involved in
concentration :-
Name:
(b) two metallic oxides which cannot be reduced by carbon, carbon monoxide or hydrogen
Explain the following terms:
(c) slag
Explain the following terms:
smelting
`ZnCO_3` ..............................
..............................
Which metal is used for:
making pipes, buckets, water tanks,
What is magnalium? Name the main elements present in it? Write its one use ?
Complete the following flow chart and answer the questions below:

i) In which method pine oil is used?
ii) Explain what method of concentration in brief.
Why does iron or zinc not occur free in nature?
Write the molecular formulae of the following compound.
Ferrous tungstate
Explain the hydraulic separation method with a neat labelled diagram.
Explain the froth floatation method with a neat labelled diagram.
Chemical formula of rust is ______
Zirconium is refined by ____________.
Name the process that is employed to refine aluminium.
Identify false statement regarding lithium aluminium hydride?
