Advertisements
Advertisements
Question
In Wilfley table method to separate particles of gangue _______ method is used.
Options
Magnetic
Froth floatation
Leaching
gravitation
Advertisements
Solution
In Wilfley table method to separate particles of gangue gravitation method is used.
APPEARS IN
RELATED QUESTIONS
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their: hydroxide
A student has been collecting silver coins and copper coins. One day she observed a black
coating on silver coins and a green coating on copper coins. Which chemical phenomenon is
responsible for these coatings? Write the names of black and green coatings.
On which factors does the purification of metals depend?
`Ag2O` ................................
................................
`MnO_2 + 4Al`  …………….
…………….
`ZnCO_3`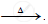 ..............................
..............................
`2Pb(NO3)_2`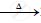 .........................
.........................
Give the ionization reactions of electrolyte used in Hall’s process. write the reaction at the cathode and the anode. Why the anode has to be replaced in this process?
what does it form with silica present in the ore?
Arrange the metals in (2) in the decreasing order of reactivity.
Explain the term of Metallurgy .
Explain with reason:
Why is powdered coke sprinkled on top of the electrolyte?
A compound that is added to lower the fusion temperature of the electrolytic bath in the extraction of aluminium.
The following is a sketch of an electrolytic cell used in the extraction of aluminium :
(a) What is the substance of which the electrode A and B are made?
(b) At which electrode (A or B) is aluminium formed?
(c) What are the two aluminium compounds in the electrolyte C?
(d) Why is it necessary for electrode B to be continuously replaced?
Write the molecular formulae of the following compound.
Copper pyrite
The chemical name of rust is ______.
State the reason for the addition of caustic alkali to bauxite ore during the purification of bauxite.
Explain the smelting process.
Calculate the difference between heat of combustion of carbon monoxide gas at constant pressure and at constant volume at 27°C? (R = 2 cal K-1 mol-1).
