Advertisements
Advertisements
Question
Hydrogen gas is diatomic whereas inert gases are monoatomic – Explain on the basis of MO theory.
Advertisements
Solution
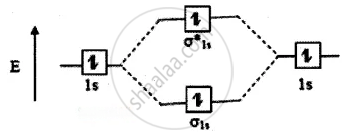
The molecular orbital electronic configuration of the hydrogen molecule is (σ1s2). The molecular orbital energy level diagram of the H2 molecule is given in

Here, N2 = 2, Na = 0
Bond order = `("N"_"b" - "N"_"a")/2`
`= (2 - 0)/2` = 1
He2: σ1s2 σ1s*2
The molecular orbital energy level diagram of He2 (hypothetical) is given in
Here, Nb = 2 and Na = 2
Bond order = `("N"_"b" - "N"_"a")/2`
`(2 - 2)/2` = 0
As the bond order for He2 comes out to form between two hydrogen atoms. But as the bond order of helium is zero, there is no bond between helium atoms and hence it is monoatomic.
Result:
As the bond order of the H2 molecule is one, it is diatomic and a single bond is formed between two hydrogen atoms. But as the bond order of helium is zero, there is no bond between helium atoms and hence it is monoatomic.
APPEARS IN
RELATED QUESTIONS
Arrange the following compounds on the basis of lattice energies in decreasing (descending) order:
BeF2, AlCl3, LiCl, CaCl2, NaCl
Give the total number of electrons around sulfur (S) in SF6 compound.
What are the interacting forces present during the formation of a molecule of a compound?
Why is O2 molecule paramagnetic?
Of the following molecules, which have shape similar to carbon dioxide?
Draw the M.O diagram for oxygen molecule calculate its bond order and show that O2 is paramagnetic.
Draw MO diagram of CO and calculate its bond order.
Of the ions Ti4+, Co2+ and Cr3+, ____________.
Which element among the following does NOT form diatomic molecules?
H2 molecule is more stable than Li2 molecule, because ______.
