Advertisements
Advertisements
प्रश्न
Hydrogen gas is diatomic whereas inert gases are monoatomic – Explain on the basis of MO theory.
Advertisements
उत्तर
The molecular orbital electronic configuration of the hydrogen molecule is (σ1s2). The molecular orbital energy level diagram of the H2 molecule is given in

Here, N2 = 2, Na = 0
Bond order = `("N"_"b" - "N"_"a")/2`
`= (2 - 0)/2` = 1
He2: σ1s2 σ1s*2
The molecular orbital energy level diagram of He2 (hypothetical) is given in
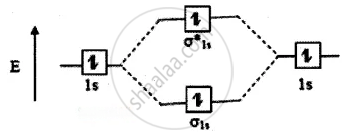
Here, Nb = 2 and Na = 2
Bond order = `("N"_"b" - "N"_"a")/2`
`(2 - 2)/2` = 0
As the bond order for He2 comes out to form between two hydrogen atoms. But as the bond order of helium is zero, there is no bond between helium atoms and hence it is monoatomic.
Result:
As the bond order of the H2 molecule is one, it is diatomic and a single bond is formed between two hydrogen atoms. But as the bond order of helium is zero, there is no bond between helium atoms and hence it is monoatomic.
APPEARS IN
संबंधित प्रश्न
Draw an orbital diagram of Hydrogen fluoride molecule
Arrange the following compounds on the basis of lattice energies in decreasing (descending) order:
BeF2, AlCl3, LiCl, CaCl2, NaCl
Assertion: Oxygen molecule is paramagnetic.
Reason: It has two unpaired electron in its bonding molecular orbital.
Pick out the incorrect statement from the following:
Of the following molecules, which have shape similar to carbon dioxide?
What do you understand by Linear combination of atomic orbitals in MO theory?
Which of the following statements is INCORRECT?
Identify the CORRECT option according to MOT.
In \[\ce{O^-2}\], O2, and \[\ce{O^{2-}2}\] molecular species, the total number of antibonding electrons, respectively, are:
According to MO theory which of the following lists ranks the nitrogen species in terms of increasing bond order?
