Advertisements
Advertisements
Question
Draw the M.O diagram for oxygen molecule calculate its bond order and show that O2 is paramagnetic.
Advertisements
Solution
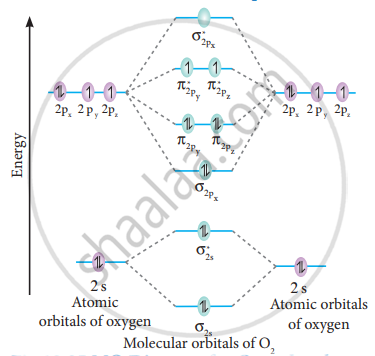
MO Diagram for O2 molecule
Electronic configuration of O atom:
1s2 2s2 2p4
Electronic configuration of O2 molecule:
σ1s2, σ1s*2, σ2s2, σ2s*2, σ2px2, π2py2 π2pz2 π2py*1 π2pz*1
Bond order = `("N"_"b" - "N"_"a")/2`
`= (10 - 6)/2`
= 2
The molecule has two unpaired electrons hence it is paramagnetic.
APPEARS IN
RELATED QUESTIONS
Arrange the following compounds on the basis of lattice energies in decreasing (descending) order:
BeF2, AlCl3, LiCl, CaCl2, NaCl
Assertion: Oxygen molecule is paramagnetic.
Reason: It has two unpaired electron in its bonding molecular orbital.
Of the following molecules, which have shape similar to carbon dioxide?
What is the bond order of B2 molecule?
Among the following MOs, the highest energy MO that is occupied by electrons in case of N2 molecule is ______.
According to MOT, the number of unpaired electrons in O2 molecule is ____________.
Identify the CORRECT option according to MOT.
Which element among the following does NOT form diatomic molecules?
The correct statement with regard to \[\ce{H^+_2}\] and \[\ce{H^-_2}\] is:
Which element among the following does not form diatomic molecules?
