Advertisements
Advertisements
प्रश्न
Draw the M.O diagram for oxygen molecule calculate its bond order and show that O2 is paramagnetic.
Advertisements
उत्तर
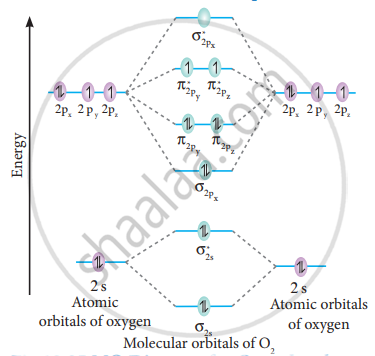
MO Diagram for O2 molecule
Electronic configuration of O atom:
1s2 2s2 2p4
Electronic configuration of O2 molecule:
σ1s2, σ1s*2, σ2s2, σ2s*2, σ2px2, π2py2 π2pz2 π2py*1 π2pz*1
Bond order = `("N"_"b" - "N"_"a")/2`
`= (10 - 6)/2`
= 2
The molecule has two unpaired electrons hence it is paramagnetic.
APPEARS IN
संबंधित प्रश्न
Select and write the most appropriate alternatives from the given choices.
Which O2 molecule is paramagnetic. It is explained on the basis of ______.
Draw an orbital diagram of Hydrogen fluoride molecule
Pick out the incorrect statement from the following:
Of the following molecules, which have shape similar to carbon dioxide?
Discuss the formation of N2 molecule using MO Theory.
Hydrogen gas is diatomic whereas inert gases are monoatomic – Explain on the basis of MO theory.
Identify the CORRECT option according to MOT.
According to molecular orbital theory, antibonding molecular orbitals of O2 contain ____________.
Which element among the following does NOT form diatomic molecules?
Which of the following molecules has bond order zero?
