Advertisements
Advertisements
Question
Draw MO diagram of CO and calculate its bond order.
Advertisements
Solution
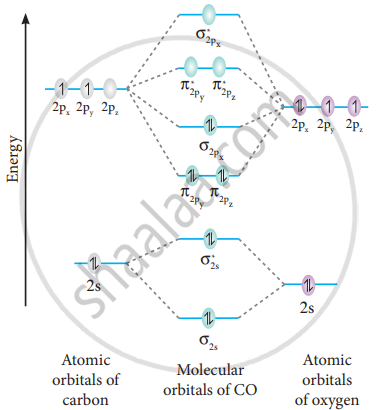
Bonding in some hetero nuclear di-atomic molecules:
Molecular orbital diagram of Carbon monoxide molecule (CO)
Electronic configuration of C atom: 1s2 2s2 2p2
Electronic configuration of O atom: 1s2 2s2 2p4
Electronic configuration of CO molecule :
σ1s2, σ1s*2, σ2s2, σ2s*2, π2py2, π2pz2 σ2px2
Bond order = `("N"_"b" - "N"_"a")/2`
`= (10 - 4)/2` = 3
The molecule has no unpaired electrons hence it is diamagnetic.
APPEARS IN
RELATED QUESTIONS
Select and write the most appropriate alternatives from the given choices.
Which of the following is true for CO2?
Select and write the most appropriate alternatives from the given choices.
Which O2 molecule is paramagnetic. It is explained on the basis of ______.
Arrange the following compounds on the basis of lattice energies in decreasing (descending) order:
BeF2, AlCl3, LiCl, CaCl2, NaCl
Give the total number of electrons around sulfur (S) in SF6 compound.
Why is O2 molecule paramagnetic?
Hydrogen gas is diatomic whereas inert gases are monoatomic – Explain on the basis of MO theory.
What is the bond order of B2 molecule?
Among the following MOs, the highest energy MO that is occupied by electrons in case of N2 molecule is ______.
The correct statement with regard to \[\ce{H^+_2}\] and \[\ce{H^-_2}\] is:
According to MO theory which of the following lists ranks the nitrogen species in terms of increasing bond order?
