Advertisements
Advertisements
Question
Give reason of Water pipes burst in severe winter.
Advertisements
Solution 1
Water pipes bursts in severe winter because the water inside I the pipes freezes and increases its volume.
Solution 2
In severe winter, water pipe lines often burst because water freeze at sub-zero temperature and in doing so expands. Since there is no space within the pipes for expanding ice, it exerts very large pressure and bursts open the steel pipes.
RELATED QUESTIONS
The amount of water vapour in air is determined in terms of its ______.
Explain the following:
In cold regions in winter, the rocks crack due to anomalous expansion of water.
1 kg of ice at 0°C is mixed with 1 kg of steam at 100°C. What will be the composition of the system when thermal equilibrium is reached? Latent heat of fusion of ice = 3.36 × 103 J kg−1 and latent heat of vaporization of water = 2.26 × 106 J kg−1.
A 50 kg man is running at a speed of 18 km h−1. If all the kinetic energy of the man can be used to increase the temperature of water from 20°C to 30°C, how much water can be heated with this energy?
Draw a graph to show the variation in density of water with temperature in the range from 0°C to 10°C.
Draw a graph between volume and temperature, when 5 cm3 of ice at -10°C is heated to form water at +10°C.
Explain, why are the exposed water pipes lagged with straw during severe winter?
Explain why do vegetables and fruits get damaged during severe frost?
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer just in contact with ice?
What are land and sea breezes? Explain with the help of a labeled diagram.
Write the name.
The instrument used to study anomalous behaviour of water.
Write a short note.
Anomalous behaviour of water
Observe the given picture and answer the following questions.
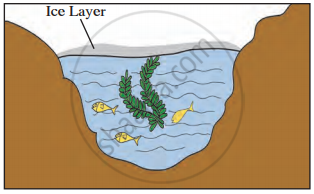
- Which property do you understand in this picture?
- What is the temperature of the water at the surface?
- What is the temperature below the layer of ice on the surface?
Match the columns:
| Column ‘A’ | Column ‘B’ |
| The density of water is maximum at | (a) 0°C |
| (b) 4°C | |
| (c) 100°C |
A graph between the volume and temperature of water is shown. Explain the anomalous behaviour of water.

