Advertisements
Advertisements
Question
Explain the term – molecules. Give three examples of atoms of the same element forming a molecule. State the atomicity of the same.
Advertisements
Solution
Atoms of the same element or different elements combine to form a molecule.
Atoms of the same element forming a molecule
1. Oxygen
2. Nitrogen
3. Hydrogen.

Atomicity is the number of atoms present in one molecule of the element.
| Element | Molecule | Atomicity |
| Hydrogen H | H2 | 2 |
| Nitrogen N | N2 | 2 |
| Oxygen O | O2 | 2 |
RELATED QUESTIONS
Write down the name of a compound represented by the following formula:
KNO3
Give the name of the element present in the following compound:
Hydrogen bromide
Give the name of the element present in the following compound:
Baking powder
Calculate the number of molecules of sulphur (S8) present in 16 g of solid sulphur.
State the term.
The number of atoms present in a molecule of an element _________
Define: Atomic number
What is the difference between the molecule of an element and the molecule of a compound ? Give one
example of each.
Molecular compounds are usually formed by the combination between :
An element A forms an oxide A2O5.
What is the valency of element A ?
An element X forms the following compounds with hydrogen, carbon and oxygen :
H2X, CX2, XO2, XO3
State the three valencies of element X which are illustrated by these compounds.
Correct the following statement:
A molecule of sulphur is monoatomic.
Why are the symbols and formulae of substances important?
Give one example of a polyatomic molecule.
State the main points of the contradiction of Dalton’s atomic theory by the Modern Atomic Theory.
Give a reason why –
Helium is considered monoatomic, but hydrogen a diatomic molecule.
State the atomicity of the following:
Neon
State the atomicity of the following:
Chlorine
State true of false. If false, give the correct statement.
NaCl represents one molecule of sodium chloride.
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 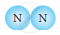 |
|
| 4. | 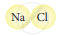 |
Which of the following is a triatomic molecule?
