Advertisements
Advertisements
प्रश्न
Explain the term – molecules. Give three examples of atoms of the same element forming a molecule. State the atomicity of the same.
Advertisements
उत्तर
Atoms of the same element or different elements combine to form a molecule.
Atoms of the same element forming a molecule
1. Oxygen
2. Nitrogen
3. Hydrogen.

Atomicity is the number of atoms present in one molecule of the element.
| Element | Molecule | Atomicity |
| Hydrogen H | H2 | 2 |
| Nitrogen N | N2 | 2 |
| Oxygen O | O2 | 2 |
संबंधित प्रश्न
Write down the name of a compound represented by the following formula:
CaCl2
Write down the name of a compound represented by the following formula:
KNO3
Give the name of the element present in the following compound:
Quick lime
Give the name of the element present in the following compound:
Baking powder
State the term.
The number of atoms present in a molecule of an element _________
Define: Atomic number
What are (i) ionic compounds, and (ii) molecular compounds ? Give two examples of each type of compounds.
An element X forms the following compounds with hydrogen, carbon and oxygen :
H2X, CX2, XO2, XO3
State the three valencies of element X which are illustrated by these compounds.
Calculate the mass of 3.011 × 1024 atoms of carbon.
State the atomicity of the following:
Oxygen
State the atomicity of the following:
Argon
Which of the following is a short and scientific way of representing one molecule of an element or compound?
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 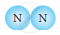 |
|
| 4. | 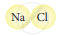 |
Two elements sometimes can form more than one compound.
Define: Atomicity.
Give any two examples for heterodiatomic molecules.
Derive the relationship between Relative molecular mass and Vapour density.
