Advertisements
Advertisements
Question
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 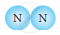 |
|
| 4. | 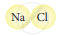 |
Answer in Brief
Advertisements
Solution
| 1. |  |
Molecule of Element |
| 2. |  |
Molecule of Compound |
| 3. |  |
Molecule of Element |
| 4. | 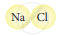 |
Molecule of Compound |
shaalaa.com
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
Write down the name of a compound represented by the following formula:
CaCl2
Give the name of the element present in the following compound:
Baking powder
Give the name of the element present in the following compound:
Potassium sulphate
Define: Mass number :
What is the atomicity of the following ?
- Oxygen
- Ozone
- Neon
- Sulphur
- Phosphorus
- Sodium
In what form do noble gases occur in nature?
Molecular compounds are usually formed by the combination between :
Give one example of a polyatomic molecule.
State the atomicity of the following:
Oxygen
Give any two examples for heterodiatomic molecules.
