Advertisements
Advertisements
Question
Explain in detail an adiabatic process.
Advertisements
Solution
Adiabatic process: This is a process in which no heat flows into or out of the system (Q = 0). But the gas can expand by spending its internal energy or gas can be compressed through some external work. So the pressure, volume and temperature of the system may change in an adiabatic process.
For an adiabatic process, the first law becomes ∆U = W.
This implies that the work is done by the gas at the expense of internal energy or work is done on the system which increases its internal energy.
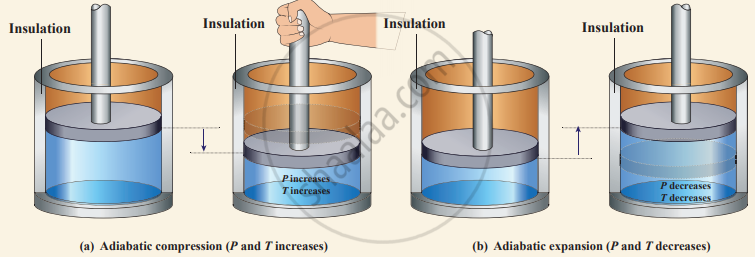
Adiabatic compression and expansion
The adiabatic process can be achieved by the following methods:
(i) Thermally insulating the system from surroundings so that no heat flows into or out of the system; for example when the thermally insulated cylinder of gas is compressed (adiabatic compression) or expanded (adiabatic expansion) as shown in the Figure.
(ii) If the process occurs so quickly that there is no time to exchange heat with surroundings even though there is no thermal insulation. A few examples are shown in Figure.
The equation of state for an adiabatic process is given by
PVγ = constant ...............(1)
Here γ is called adiabatic exponent `(γ = "C"_"p"/"C"_"v")` which depends on the nature of the gas. The equation (1) implies that if the gas goes from an equilibrium state (Pi, Vi) to another equilibrium state (Pf, Vf) adiabatically then it satisfies the relation
`"P"_"i""V"_"i"^γ = "P"_"f""V"_"f"^γ` ..........(2)
The PV diagram for an adiabatic process is also called adiabat. But actually, the adiabatic curve is steeper than an isothermal curve.
We can also rewrite the equation (1) in terms of T and V. From the ideal gas equation, the pressure P = `(μ"RT")/"V"`.
Substituting this equation in equation (1), we have
`(μ"RT")/"V""V^γ` = constant (or) `"T"/"V""V"^γ = "constant"/(μ"R")`
Note here that is another constant. So it can be written as
TVγ-1 = constant ..................(3)
The equation implies that if the gas goes from an initial equilibrium state (Ti, Vi) to final equilibrium state (Tf, Vf) adiabatically then it satisfies the relation
`"T"_"i""V"_"i"^(γ - 1) = "T"_"f""V"_"f"^(γ - 1)` .......(4)
The equation of state for the adiabatic process can also be written in terms of T and P as
TγP1-γ = constant ............(5)

PV diagram for adiabatic expansion and adiabatic compression
APPEARS IN
RELATED QUESTIONS
Draw a p-V diagram of the reversible process.
State the assumptions made for thermodynamic processes.
Differentiate between the reversible and irreversible processes.
Draw the PV diagram for the isobaric process.
Explain in detail the isochoric process.
In an isothermal process, the volume of an ideal gas is halved. One can say that ____________.
We consider a thermodynamic system. If `Delta"U"` represents the increase in its internal energy and W the work done by the system, which of the following statements is true?
Which of the following processes is reversible?
Give any two types of a thermodynamic process.
In a certain thermodynamical process, the pressure of a gas depends on its volume as kV3. The work done when the temperature changes from 100°C to 300°C will be ______ nR, where n denotes number of moles of a gas.
