Advertisements
Advertisements
Question
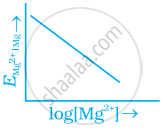
Electrode potential for Mg electrode varies according to the equation
`E_(Mg^(2+) | Mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log 1/([Mg^(2+)])`. The graph of `E_(Mg^(2+) | Mg)` vs `log [Mg^(2+)]` is ______.
Options
Advertisements
Solution
Electrode potential for Mg electrode varies according to the equation
`E_(Mg^(2+) | Mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log 1/([Mg^(2+)])`. The graph of `E_(Mg^(2+) | Mg)` vs `log [Mg^(2+)]` is a straignt line with a positive slope and intercept `E_((Mg^(2+))/(Mg)`.

Explanation:
`E_(Mg^(2+) | mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log [Mg^(2+)]`
Compare this equation with the equation of straingt line y = mx + c.
The graph of `E_(Mg^(2+) | Mg)` versus `log [Mg^(2+)]` is a straignt line with a positive slope and intercept `E_((Mg^(2+))/(Mg)`.
APPEARS IN
RELATED QUESTIONS
Write cathode and anode reaction in a fuel cell.
If 'I' stands for the distance between the electrodes and 'a' stands for the area of cross-section of the electrode, `"l"/"a"` refers to ____________.
At 25°C, the emf of the following electrochemical cell.
\[\ce{Ag_{(s)} | Ag^+ (0.01 M) | | Zn^{2+} {(0.1 M)} | Zn_{(s)}}\] will be:
(Given \[\ce{E^0_{cell}}\] = −1.562 V)
How many faradays of electricity are required for the following reaction to occur
\[\ce{MnO^-_4 -> Mn^2+}\]
Cell equation: \[\ce{A + 2B^- -> A^{2+} + 2B}\]
\[\ce{A^{2+} + 2e^- -> A}\] E0 = +0.34 V and log10 k = 15.6 at 300 K for cell reactions find E0 for \[\ce{B^+ + e^- -> B}\]
Define anode
Describe the electrolysis of molten NaCl using inert electrodes.
Why is anode in galvanic cell considered to be negative and cathode positive electrode?
Write a note on sacrificial protection.
Which of the following statement is correct?
Use the data given in below find out the most stable oxidised species.
`E^0 (Cr_2O_1^(2-))/(Cr_(3+))` = 1.33 V `E^0 (Cl_2)/(Cl^-)` = 1.36 V
`E^0 (MnO_4^-)/(MN^(2+))` = 1.51 V `E^0 (Cr^(3+))/(Cr)` = – 0.74 V
For the given cell, \[\ce{Mg | Mg^{2+} || Cu^{2+} | Cu}\]
(i) \[\ce{Mg}\] is cathode
(ii) \[\ce{Cu}\] is cathode
(iii) The cell reaction is \[\ce{Mg^+ Cu^{2+} -> Mg^{2+} + Cu}\]
(iv) \[\ce{Cu}\] is the oxidising agent
Consider the following diagram in which an electrochemical cell is coupled to an electrolytic cell. What will be the polarity of electrodes ‘A’ and ‘B’ in the electrolytic cell?
A galvanic cell has electrical potential of 1.1V. If an opposing potential of 1.1V is applied to this cell, what will happen to the cell reaction and current flowing through the cell?
Match the items of Column I and Column II.
| Column I | Column II |
| (i) K | (a) I × t |
| (ii) Λm | (b) `Λ_m/Λ_m^0` |
| (iii) α | (c) `K/c` |
| (iv) Q | (d) `G^∗/R` |
If the half-cell reaction A + e– → A– has a large negative reduction potential, it follow that:-
Galvanic cell is a device in which ______.
What should be the signs (positive/negative) for \[\ce{E^0_{cell}}\] and ΔG0 for a spontaneous redox reaction occurring under standard conditions?
Can we construct an electrochemical cell with two half-cells composed of ZnSO4 solution and zinc electrodes? Explain your answer.
The cell constant of a conductivity cell is 0.146 cm-1. What is the conductivity of 0.01 M solution of an electrolyte at 298 K, if the resistance of the cell is 1000 ohm?