Advertisements
Advertisements
Question
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Lithium [At. no. = 3, Mass no. = 7]
Advertisements
Solution
Structure of atoms:
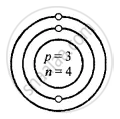
Z is Atomic Number A is the mass number
Lithium \[\ce{^7_3Li}\]
Z = 3 = p = e
K L
e = 3 = 2, 1
A = p + n
7 = 3 + 3
∴ n = 7 − 3 = 4
APPEARS IN
RELATED QUESTIONS
Explain the Atomic number with example.
Write true or false for the following statement
An atom on the whole has a positive charge.
What are the two main features of Rutherford’s atomic model?
The properties of the elements are a periodic function of their __________.
The atomic number and mass number of sodium are 11 and 23 respectively. What information is conveyed by this statement?
An element A atomic number 7 mass numbers 14
B electronic configuration 2,8,8
C electrons 13, neutrons 14
D Protons 18 neutrons 22
E Electronic configuration 2,8,8,1
State Valency of each element
An element A atomic number 7 mass numbers 14
B electronic configuration 2,8,8
C electrons 13, neutrons 14
D Protons 18 neutrons 22
E Electronic configuration 2,8,8,1
State (i) Valency of each element (ii) which one is a metal (iii) which is non-metal (iv) which is an inert gas
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Oxygen – \[\ce{^16_8O}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Which sub-atomic particles in the 3 isotopes of ‘H’ are the same.
Differentiate mass number from atomic number.
What are nucleons? How many nucleons are present in Phosphorous? Draw its structure.
