Advertisements
Advertisements
प्रश्न
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Lithium [At. no. = 3, Mass no. = 7]
Advertisements
उत्तर
Structure of atoms:
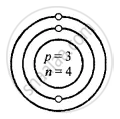
Z is Atomic Number A is the mass number
Lithium \[\ce{^7_3Li}\]
Z = 3 = p = e
K L
e = 3 = 2, 1
A = p + n
7 = 3 + 3
∴ n = 7 − 3 = 4
APPEARS IN
संबंधित प्रश्न
Explain the Mass number with example.
Complete the following table.
| Atomic number | Mass number | Number of Neutrons | Number of protons | Number of electrons | Name of the Atomic species |
| 9 | - | 10 | - | - | - |
| 16 | 32 | - | - | - | Sulphur |
| - | 24 | - | 12 | - | - |
| - | 2 | - | 1 | - | - |
| - | 1 | 0 | 1 | 0 | - |
Give the following a suitable word/phrase.
Atoms of same element with same atomic number but a different mass number.
Multiple Choice Questions
The sub atomic particle with negative charge is
State why the atomic weight of an element is also termed relative atomic mass.
Differentiate between the following term:
Atomic number and mass number
Complete the following table.
| Atomic Number | Mass Number | Number of Neutrons | Number of Protons | Number of Electrons | Name of the Element |
| 9 | ______ | 10 | ______ | ______ | ______ |
| 16 | ______ | 16 | ______ | ______ | ______ |
| ______ | 24 | ______ | ______ | 12 | Magnesium |
| ______ | 2 | ______ | 1 | ______ | ______ |
| ______ | 1 | 0 | 1 | 1 | ______ |
Give short answer:
what is the atomic mass number? mention its formula.
K and L shells can accommodate a maximum of 2 and 8 electrons respectively.
What are the letters A, Z, and X stand for?
