Advertisements
Advertisements
प्रश्न
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Lithium [At. no. = 3, Mass no. = 7]
Advertisements
उत्तर
Structure of atoms:
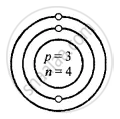
Z is Atomic Number A is the mass number
Lithium \[\ce{^7_3Li}\]
Z = 3 = p = e
K L
e = 3 = 2, 1
A = p + n
7 = 3 + 3
∴ n = 7 − 3 = 4
APPEARS IN
संबंधित प्रश्न
Dalton said that ______ could not be divided.
Fill in the blanks.
Isotopes are the atoms of ______ element having the ______ atomic number but ______ mass number.
Multiple Choice Questions
The sub atomic particle with negative charge is
FILL IN THE BLANK
Rutherford won the Nobel Prize in 1908 for his investigations in the field of ......................
Fill in the blank of the following statement :
The total number of protons and neutrons in the nucleus of an atom is called its _________.
An atom of an element has two electrons in the M shell.
What is the (a) atomic number (b) number of protons in this element?
Give short answer:
what is the atomic mass number? mention its formula.
A ______ number describes a specific aspect of an electron.
Mass No =______ + No. of Neutrons.
An element has 2 electrons in its N shell.
What is its atomic number?
