Advertisements
Advertisements
Questions
Draw optical isomers of [Co(en)3]3+.
Write the structures of the optical isomers of [Co(en)3]3+.
Advertisements
Solution
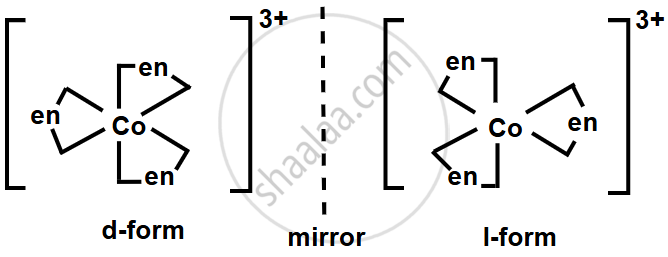
RELATED QUESTIONS
Write the structural formula and IUPAC names of all possible isomers of the compound with molecular formula C3H8O.
Give one chemical test as an evidence to show that [Co (NH3)5Cl] are ionisation isomers.
Answer in brief.
What are ionization isomers ? Give an example.
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)3]4⊕
Define the term Hydrated isomers.
Draw structure of cis isomer of [Co(NH3)4Cl2]+
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
Fac-mer isomerism is shown by
What is linkage isomerism? Explain with an example.
Which of the following is NOT a pair of structural isomers?
Identify the CORRECT statement about the following complex of platinum.
[PtCl2(en)2]2+
The number of geometrical isomers of [CrCl2(en)2]+ is ____________.
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
Which of the following has zero dipole moment?
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
\[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2 Cl(NO2)]}\] is ______.
Assertion: Complexes of MX6 and MX5L type (X and L are unidentate) do not show geometrical isomerism.
Reason: Geometrical isomerism is not shown by complexes of coordination number 6.
The correct structure of Fe(CO)5 is
Which of the following does not show optical isomerism?
Which among the following solid is a non-polar solid?
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Draw the structure of cis isomers of Pt(NH3)2Cl2.
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Name the type of isomerism exhibited by the following pair of compounds:
[Cr(H2O)5Cl]Cl2.H2O and [Cr(H2O)4Cl2]Cl.2H2O
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Pt(NH3)4Cl2]Br2 and [Pt(NH3)4 Br2]Cl2}\]
Match List-I with List-II:
| List-I (Complex) |
List-II (Type of isomerism) |
| A. [Co(NH3)5(NO2)]Cl2 | I. Solvate isomerism |
| B. [Co(NH3)5(SO4)]Br | II. Linkage isomerism |
| C. [Co(NH3)6][Cr(CN)6] | III. Ionisation isomerism |
| D. [Co(H2O)6]Cl3 | IV. Coordination isomerism |
Choose the correct answer from the options given below:
