Advertisements
Advertisements
Question
Distinguish between Inductive effect and resonance effect.
Advertisements
Solution
| Inductive effect | Resonance effect | |
| 1. | The presence of a polar covalent bond is required. | The presence of a conjugated π electron system or species having an atom carrying p orbital attached to multiple bonds is required. |
| 2. | The polarity is induced in adjacent carbon-carbon single (covalent) bonds due to the presence of an influencing group (more electronegative atom than carbon). | The polarity is produced in the molecule by the interaction of conjugated π bonds (or that between π bond and p orbital on the adjacent atom). |
| 3. | Depending on the nature of the influencing group it is differentiated as +I effect and -I effect. | Depending on the nature of the influencing group it is differentiated as +R and -R effect. |
| 4. | The direction of the arrow head denotes the direction of the permanent electron displacement. | The delocalisation of π electrons is denoted by using curved arrows. |
APPEARS IN
RELATED QUESTIONS
Find out the most stable species from the following. Justify
`dot"CH","CH"_3-dot"CH" - "CH"_3,` \[\begin{array}{cc}\ce{CH3 -\dot{C} - CH3}\\
|\phantom{.}\\\phantom{..}\ce{CH3}\end{array}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.

Identify the α-carbon in the following species and give the total number of α-hydrogens.
\[\ce{CH2 = CH - CH2 - CH3}\]
Draw a resonance structure of the following:
Benzaldehyde
Draw a resonance structure of the following:
Acetate ion
Write true or false. Correct the false statement.
Homolytic fission involves the unsymmetrical breaking of a covalent bond.
Write true or false. Correct the false statement.
Heterolytic fission results in the formation of free radicals.
Write true or false. Correct the false statement.
Free radicals are negatively charged species.
Write true or false. Correct the false statement.
Aniline is a heterocyclic compound.
Choose the correct option.
Which of the following statements are true with respect to electronic displacement in a covalent bond?
a. Inductive effect operates through π bond
b. Resonance effect operates through σ bond
c. Inductive effect operates through σ bond
d. Resonance effect operates through π bond
Choose the correct option.
Hyperconjugation involves overlap of ______ orbitals.
The correct IUPAC name of the compound 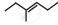 is ______.
is ______.
The best reagent for the following conversion is:

Which of the following represents a set of nucleophiles?
Which of the following is NOT an electrophile?
How many pi bonds and sigma bonds are present in following molecule?
The most probable product in the reaction given below is:

Which element among the following does form pπ - pπ multiple bonds?
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H -CH2 - CH2 }\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H - CH2 - CH3}\]
Identify the α - carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H -CH2 - CH3}\]
Identify the α - carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Which of the following groups exhibits a +I effect?
