Advertisements
Advertisements
Question
Calculate the oxidation number of sulphur, chromium and nitrogen in H2SO5, `"Cr"_2"O"_7^(2-)` and `"NO"_3^-`. Suggest structure of these compounds. Count for the fallacy.
Advertisements
Solution
1)
+1 x -2
\[\ce{H2SO5}\]
2(+1) + 1(x) + 5(-2) = 0
⇒ 2 + x - 10 = 0
⇒ x = + 8
However, the O.N. of S cannot be +8. S has six valence electrons. Therefore, the O.N. of S cannot be more than +6.
The structure of H2SO5 is shown as follows:
\[\begin{array}{cc}
\ce{^{-2}O}\phantom{...........}\\
||\phantom{.........}\\
\ce{^{+1}H - ^{-2}O - S^{\text{x}} - ^{-1}O - ^{-1}O - ^{+1}H}\\
||\phantom{.........}\\
\ce{^{-2}O}\phantom{............}\end{array}\]
Now, 2(+1) + 1(x) + 3(-2) + 2(-1) = 0
⇒ 2 + x - 6 - 2 = 0
⇒ x = +6
Therefore, the O.N. of S is +6.
2)
x 2-
\[\ce{Cr2 O^{2-}_7}\]
2(x) + 7(-2) = -2
⇒ 2x - 14 = -2
⇒ x = +6
Here, there is no fallacy about the O.N. of Cr in `"Cr"_2"O"_7^(2-)`.
The structure of `"Cr"_2"O"_7^(2-)` is shown as follows:
\[\begin{array}{cc}
\ce{^{2-}O}\phantom{...........}\ce{^{2-}O}\phantom{....}\\
||\phantom{..............}||\phantom{..}\\
\ce{^{2-}O = Cr^{+6} - ^{2-}O - Cr^{+6} - O^{2-}}\\
|\phantom{..............}|\phantom{...}\\
\ce{_{1-}O^-}\phantom{.........}\ce{_{1-}O^-}\phantom{....}\end{array}\]
Here, each of the two Cr atoms exhibits the O.N. of +6.
3)
x 2-
\[\ce{N O^-_3}\]
1(x) + 3(-2) = -1
⇒ x - 6 = -1
⇒ x = +5
Here, there is no fallacy about the O.N. of N in `"NO"_3^(-)`
The structure of `"NO"_3^(-)` is shown as follows
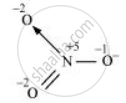
The N atom exhibits the O.N. of +5.
APPEARS IN
RELATED QUESTIONS
Consider the reaction:
\[\ce{O3(g) + H2O2(l) → H2O(l) + 2O2(g)}\]
Why it is more appropriate to write these reaction as:
\[\ce{O3(g) + H2O2 (l) → H2O(l) + O2(g) + O2(g)}\]
Also, suggest a technique to investigate the path of the redox reactions.
How do you count for the following observations?
Though alkaline potassium permanganate and acidic potassium permanganate both are used as oxidants, yet in the manufacture of benzoic acid from toluene we use alcoholic potassium permanganate as an oxidant. Why? Write a balanced redox equation for the reaction.
Balance the following equation in basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{Cl_2O_{7(g)} + H_2O_{2(aq)} -> ClO-_{2(aq)} + O_{2(g)} + H+_{(aq)}}\]
The Mn3+ ion is unstable in solution and undergoes disproportionation to give Mn2+, MnO2, and H+ ion. Write a balanced ionic equation for the reaction.
Justify that the following reaction is redox reaction; identify the species oxidized/reduced, which acts as an oxidant and which acts as a reductant.
\[\ce{2Cu2O_{(S)} + Cu2S_{(S)}->6Cu_{(S)} + SO2_{(g)}}\]
Balance the following redox equation by half-reaction method.
\[\ce{H2C2O_{4(aq)} + MnO^-_{4(aq)}->CO2_{(g)} + Mn^2+_{( aq)}(acidic)}\]
Identify coefficients 'x' and 'y' for the following reaction.
\[\ce{{x}H2O2_{(aq)} + ClO^-_{4(aq)} -> 2O2_{(g)} + ClO^-_{2(aq)} + {y}H2O_{(l)}}\]
Identify the oxidising agent in the following reaction:
\[\ce{CH4_{(g)} + 2O2_{(g)} -> CO2_{(g)} + 2H2O_{(l)}}\]
When methane is burnt completely, oxidation state of carbon changes from ______.
Write balanced chemical equation for the following reactions:
Dichlorine heptaoxide \[\ce{(Cl2O7)}\] in gaseous state combines with an aqueous solution of hydrogen peroxide in acidic medium to give chlorite ion \[\ce{(ClO^{-}2)}\] and oxygen gas. (Balance by ion-electron method)
Balance the following equations by the oxidation number method.
\[\ce{Fe^{2+} + H^{+} + Cr2O^{2-}7 -> Cr^{3+} + Fe^{3+} + H2O}\]
Balance the following equations by the oxidation number method.
\[\ce{I2 + S2O^{2-}3 -> I- + S4O^{2-}6}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{Fe2O3 (s) + 3CO (g) ->[Δ] 2Fe (s) + 3CO2 (g)}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{PCl3 (l) + 3H2O (l) -> 3HCl (aq) + H3PO3 (aq)}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + SO^{2-}3 + H^{+} -> Mn^{2+} + SO^{2-}4 + H2O}\]
In acidic medium, reaction, \[\ce{MNO^-_4 → Mn^2+}\] an example of ____________.
In \[\ce{Cu^{2+} + Ag -> Cu + Ag^+}\], oxidation half-reaction is:
Consider the following reaction:
\[\ce{xMnO^-_4 + yC2O^{2-}_4 + zH^+ -> xMn^{2+} + 2{y}CO2 + z/2H2O}\]
The values of x, y, and z in the reaction are, respectively:
