Advertisements
Advertisements
Question
Balance the following equation by the oxidation number method.
\[\ce{Cu + HNO3 -> Cu(NO3)2 + NO2 + H2O}\]
Advertisements
Solution
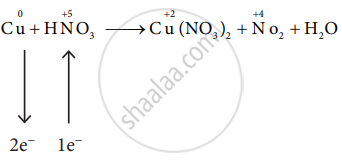
\[\ce{Cu + 2HNO3 -> Cu(NO3)2 + NO2 + H2O}\]
\[\ce{Cu + 2HNO3 + 2HNO3 -> Cu(NO3)2 + 2NO2 + 2H2O}\]
\[\ce{Cu + 4HNO3 -> Cu(NO3)2 + 2NO2 + 2H2O}\]
APPEARS IN
RELATED QUESTIONS
1 g of an impure sample of magnesium carbonate (containing no thermally decomposable impurities) on complete thermal decomposition gave 0.44 g of carbon dioxide gas. The percentage of impurity in the sample is
Total number of electrons present in 1.7 g of ammonia is
Which of the following contains the same number of carbon atoms as in 6 g of carbon-12.
What do you understand by the term oxidation number?
Balance the following equation by the ion electron method.
\[\ce{C2O^2-_4 + Cr2O^2-_7 -> Cr^3+ + CO2}\] (in acid medium)
Balance the following equation by the ion electron method.
\[\ce{Na2S2O3 + I2 -> Na2S4O6 + NaI}\]
Balance the following equation by the ion electron method.
\[\ce{Zn + NO^-_3 ->Zn^2+ + NO}\] (in acid medium)
Which of the following reagents is used for the following conversion?
\[\ce{CH3 - CH = CH - CHO -> CH3 - CH = CH - CH2OH}\]
In the redox reaction, \[\ce{2MnO^-_4 + SO^{2-}_3 + H2O -> 2MnO^{2-}_4 + SO^{2-}_4 + 2H^+}\], the reductant is:
In the cell represented by \[\ce{Pb(s) | Pb^{2+} (1 M) || Ag+ (1 M) | Ag(s)}\], the reducing agent is ______.
