Advertisements
Advertisements
प्रश्न
Balance the following equation by the oxidation number method.
\[\ce{Cu + HNO3 -> Cu(NO3)2 + NO2 + H2O}\]
Advertisements
उत्तर
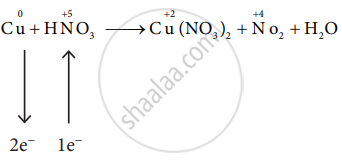
\[\ce{Cu + 2HNO3 -> Cu(NO3)2 + NO2 + H2O}\]
\[\ce{Cu + 2HNO3 + 2HNO3 -> Cu(NO3)2 + 2NO2 + 2H2O}\]
\[\ce{Cu + 4HNO3 -> Cu(NO3)2 + 2NO2 + 2H2O}\]
APPEARS IN
संबंधित प्रश्न
1 g of an impure sample of magnesium carbonate (containing no thermally decomposable impurities) on complete thermal decomposition gave 0.44 g of carbon dioxide gas. The percentage of impurity in the sample is
When 6.3 g of sodium bicarbonate is added to 30 g of acetic acid solution, the residual solution is found to weigh 33 g. The number of moles of carbon dioxide released in the reaction is
Choose the disproportionation reaction among the following redox reactions.
The correct increasing order of the oxidation state of sulphur in the anions \[\ce{SO^2-_4, SO^2-_3, S2O^2-_4, S2O^2-_6}\] is
Hydrogen peroxide is an oxidising agent. It oxidises ferrous ion to ferric ion and reduced itself to water. Write a balanced equation.
Balance the following equation by the oxidation number method.
\[\ce{KMnO4 + H2C2O4 + H2SO4 -> K2SO4 + MnSO4 + CO2 + H2O}\]
Which of the following reagents is used for the following conversion?
\[\ce{CH3 - CH = CH - CHO -> CH3 - CH = CH - CH2OH}\]
Which one of the following CANNOT act as a reducing agent?
In the cell represented by \[\ce{Pb(s) | Pb^{2+} (1 M) || Ag+ (1 M) | Ag(s)}\], the reducing agent is ______.
Which is the best oxidizing agent among the following?
