Advertisements
Advertisements
प्रश्न
Balance the following equation by the oxidation number method.
\[\ce{KMnO4 + H2C2O4 + H2SO4 -> K2SO4 + MnSO4 + CO2 + H2O}\]
Advertisements
उत्तर
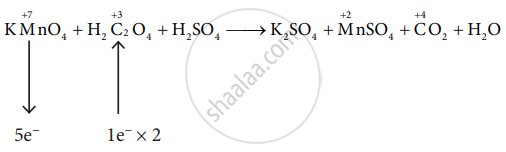
\[\ce{2KMnO4 + 5H2C2O4 + H2SO4 -> K2SO4 + MnSO4 + CO2 + H2O}\]
\[\ce{2KMnO4 + 5H2C2O4 + H2SO4 -> K2SO4 + 2MnSO4 + 10CO2 + H2O}\]
\[\ce{2KMnO4 + 5H2C2O4 + 3H2SO4 -> K2SO4 + 2MnSO4 + 10CO2 + 8H2O}\]
APPEARS IN
संबंधित प्रश्न
When 6.3 g of sodium bicarbonate is added to 30 g of acetic acid solution, the residual solution is found to weigh 33 g. The number of moles of carbon dioxide released in the reaction is
The correct increasing order of the oxidation state of sulphur in the anions \[\ce{SO^2-_4, SO^2-_3, S2O^2-_4, S2O^2-_6}\] is
Which of the following contains the same number of carbon atoms as in 6 g of carbon-12.
Balance the following equation by the oxidation number method.
\[\ce{K2Cr2O7 + Kl + H2SO4 -> K2SO4 + Cr2(SO4)3 + I2 + H2O}\]
Balance the following equation by the oxidation number method.
\[\ce{KMnO4 + Na2SO3 -> MnO2 + Na2SO4 + KOH}\]
Balance the following equation by the oxidation number method.
\[\ce{Cu + HNO3 -> Cu(NO3)2 + NO2 + H2O}\]
Balance the following equation by the ion electron method.
\[\ce{KMnO4 + SnCl2 + HCl -> MnCl2 + SnCl4 + H2O + KCl}\]
Balance the following equation by the ion electron method.
\[\ce{C2O^2-_4 + Cr2O^2-_7 -> Cr^3+ + CO2}\] (in acid medium)
Balance the following equation by the ion electron method.
\[\ce{Na2S2O3 + I2 -> Na2S4O6 + NaI}\]
What is the formula of hydrolith?
