Advertisements
Advertisements
प्रश्न
Hydrogen peroxide is an oxidising agent. It oxidises ferrous ion to ferric ion and reduced itself to water. Write a balanced equation.
Advertisements
उत्तर
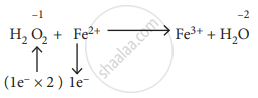
\[\ce{1H2O2 + 2Fe^2+ -> Fe^3+H2O}\]
\[\ce{H2O2 + 2Fe^2+ + 2H^+ -> 2Fe^3+ + 2H2O}\]
APPEARS IN
संबंधित प्रश्न
When 6.3 g of sodium bicarbonate is added to 30 g of acetic acid solution, the residual solution is found to weigh 33 g. The number of moles of carbon dioxide released in the reaction is
What do you understand by the term oxidation number?
Balance the following equation by the oxidation number method.
\[\ce{K2Cr2O7 + Kl + H2SO4 -> K2SO4 + Cr2(SO4)3 + I2 + H2O}\]
Balance the following equation by the oxidation number method.
\[\ce{KMnO4 + Na2SO3 -> MnO2 + Na2SO4 + KOH}\]
Balance the following equation by the ion electron method.
\[\ce{KMnO4 + SnCl2 + HCl -> MnCl2 + SnCl4 + H2O + KCl}\]
Balance the following equation by the ion electron method.
\[\ce{C2O^2-_4 + Cr2O^2-_7 -> Cr^3+ + CO2}\] (in acid medium)
Balance the following equation by the ion electron method.
\[\ce{Na2S2O3 + I2 -> Na2S4O6 + NaI}\]
Reducing agent is a species that ____________.
Which of the following reagents is used for the following conversion?
\[\ce{CH3 - CH = CH - CHO -> CH3 - CH = CH - CH2OH}\]
What is the formula of hydrolith?
