Advertisements
Advertisements
Question
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
Options
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct statement but reason is wrong statement.
Assertion is wrong statement but reason is correct statement.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Advertisements
Solution
Assertion is wrong statement but reason is correct statement.
Explanation:
Bond angle in ethers is slightly more than the tetrahedral angle due to repulsion between two bulky alkyl group.
APPEARS IN
RELATED QUESTIONS
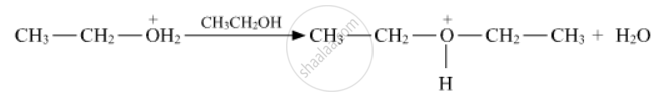
Write the mechanism of the following reaction :

Give the equation of the following reaction:
Dilute HNO3 with phenol.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas reagent is ____________.
The compound which reacts fastest with Lucas reagent at room temperature is:
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Lucas test is done to differentiate between ____________.
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
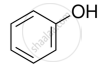
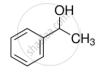
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
What is Lucas reagent?
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
