Advertisements
Advertisements
Question
A heavy nucleus X of mass number 240 and binding energy per nucleon 7.6 MeV is split into two fragments Y and Z of mass numbers 110 and 130. The binding energy of nucleons in Y and Z is 8.5 MeV per nucleon. Calculate the energy Q released per fission in MeV.
Advertisements
Solution
Total energy of nucleus X = 240 × 7.6 = 1824 MeV
Total energy of nucleus Y = 110 × 8.5 = 935 MeV
Total energy of nucleus Z = 130 × 8.5 = 1105 MeV
Therefore, energy released from fission, Q = 935 + 1105 − 1824 = 216 MeV
APPEARS IN
RELATED QUESTIONS
Write the relationship between the size of a nucleus and its mass number (A)?
Using the curve for the binding energy per nucleon as a function of mass number A, state clearly how the release in energy in the processes of nuclear fission and nuclear fusion can be explained.
As the mass number A increases, the binding energy per nucleon in a nucleus
Which of the following is a wrong description of binding energy of a nucleus?
In one average-life,
For nuclei with A > 100,
(a) the binding energy of the nucleus decreases on an average as A increases
(b) the binding energy per nucleon decreases on an average as A increases
(c) if the nucleus breaks into two roughly equal parts, energy is released
(d) if two nuclei fuse to form a bigger nucleus, energy is released.
Calculate the mass of an α-particle. Its Its binding energy is 28.2 MeV.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
What is the unit of mass when measured on the atomic scale?
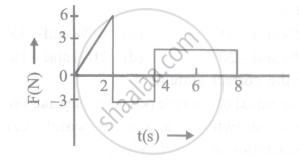
The force 'F' acting on a particle of mass 'm' is indicated by the force-time graph shown below. The change in momentum of the particle over the time interval from zero to 8s is:
The ratio of wavelengths of the last line of Balmer series and the last line of Lyman series is:
