Advertisements
Advertisements
Question
Using the curve for the binding energy per nucleon as a function of mass number A, state clearly how the release in energy in the processes of nuclear fission and nuclear fusion can be explained.
Advertisements
Solution

The rising of the binding energy curve at low mass numbers, indicates that energy will be released if two nuclides of small mass number combine to form a single middle-mass nuclide. This process is called nuclear fusion.
Highest peak (Fe) represents the most stable nucleus and all the other nuclei tend to achieve this state by undergoing nuclear reaction.
The eventual dropping of the binding energy curve at high mass numbers indicates that nucleons are more tightly bound when they are assembled into two middle-mass nuclides rather than into a single high-mass nuclide. Hence energy can be released by the nuclear fission, or splitting, of a single massive nucleus into two smaller fragments.
APPEARS IN
RELATED QUESTIONS
Asha's mother read an article in the newspaper about a disaster that took place at Chernobyl. She could not understand much from the articles and asked a few questions from Asha regarding the article. Asha tried to answer her mother's questions based on what she learnt in Class XII Physics.
(a) What was the installation at Chernobyl where the disaster took place? What according to you, was the cause of this disaster?
(b) Explain the process of release of energy in the installation at Chernobyl.
(c) What according to you, were the values displayed by Asha and her mother?
In a typical nuclear reaction, e.g.
`"_1^2H+"_1^2H ->"_2^3He + n + 3.27 \text { MeV },`
although number of nucleons is conserved, yet energy is released. How? Explain.
Suppose we have 12 protons and 12 neutrons. We can assemble them to form either a 24Mg nucleus or two 12C nuclei. In which of the two cases more energy will be liberated?
In one average-life,
For nuclei with A > 100,
(a) the binding energy of the nucleus decreases on an average as A increases
(b) the binding energy per nucleon decreases on an average as A increases
(c) if the nucleus breaks into two roughly equal parts, energy is released
(d) if two nuclei fuse to form a bigger nucleus, energy is released.
Calculate the mass of an α-particle. Its Its binding energy is 28.2 MeV.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
(a) Calculate the energy released if 238U emits an α-particle. (b) Calculate the energy to be supplied to 238U it two protons and two neutrons are to be emitted one by one. The atomic masses of 238U, 234Th and 4He are 238.0508 u, 234.04363 u and 4.00260 u respectively.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
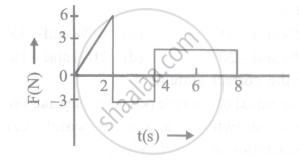
The force 'F' acting on a particle of mass 'm' is indicated by the force-time graph shown below. The change in momentum of the particle over the time interval from zero to 8s is:
A nucleus of mass M emits a γ-ray photon of frequency 'v'. The loss of internal energy by the nucleus is ______.
The ratio of wavelengths of the last line of Balmer series and the last line of Lyman series is:
