Advertisements
Advertisements
Question
A Carnot engine whose efficiency is 45% takes heat from a source maintained at a temperature of 327°C. To have an engine of efficiency of 60% what must be the intake temperature for the same exhaust (sink) temperature?
Advertisements
Solution
Efficiency of Carnot engine (η1) = 45% = 0.45
Initial intake temperature (T1) = 327°C = 600 K
New efficiency (η2) = 60% = 0.6
Efficiency of Carnot engine is given by
η = `1 - "T"_2/"T"_1`
T1 is the temperature of the source; T2 is the temperature of the sink
1st Case: T2 = (1 − η)T1 = (1 − 0.45) × 600
T2 = 330 K
2nd Case: `"T"_2/"T"_1 = 1 - η`
T1 = `"T"_2/(1 - η)`
= `330/(1 - 0.6)`
= `330/0.4`
T1 = 825 K
= 825 − 273
T1 = 552°C
APPEARS IN
RELATED QUESTIONS
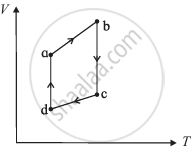
The figure shows the V-T diagram for one cycle of a hypothetical heat engine which uses the ideal gas. Draw the p-V diagram diagram of the system.
The efficiency of a heat engine working between the freezing point and boiling point of water is ____________.
State Kelvin-Planck's statement of the second law of thermodynamics.
State the second law of thermodynamics in terms of entropy.
Explain the heat engine and obtain its efficiency.
Explain in detail the Carnot heat engine.
Explain the second law of thermodynamics in terms of entropy.
Suppose a person wants to increase the efficiency of the reversible heat engine that is operating between 100°C and 300°C. He had two ways to increase efficiency.
- By decreasing the cold reservoir temperature from 100°C to 50°C and keeping the hot reservoir temperature constant
- by increasing the temperature of the hot reservoir from 300°C to 350°C by keeping the cold reservoir temperature constant.
Which is the suitable method?
A heat engine operates between a cold reservoir at temperature T2 = 400 K and a hot reservoir at temperature T1. It takes 300 J of heat from the hot re ervoir and delivers 240 J of heat to the cold reservoir in a cycle. The minimum temperature of the hot reservoir has to be ______ K.
What does a heat engine consist of?
