Advertisements
Advertisements
Question
State the second law of thermodynamics in terms of entropy.
Advertisements
Solution
“For all the processes that occur in nature (irreversible process), the entropy always increases. For reversible process entropy will not change”. Entropy determines the direction in which natural processes should occur.
APPEARS IN
RELATED QUESTIONS
A gas contained in a cylinder surrounded by a thick layer of insulating material is quickly compressed has there been a transfer of heat?
Answer in brief.
What sets the limits on the efficiency of a heat engine?
State Clausius form of the second law of thermodynamics.
Define heat engine.
Why does heat flow from a hot object to a cold object?
Explain the heat engine and obtain its efficiency.
Derive the expression for Carnot engine efficiency.
Suppose a person wants to increase the efficiency of the reversible heat engine that is operating between 100°C and 300°C. He had two ways to increase efficiency.
- By decreasing the cold reservoir temperature from 100°C to 50°C and keeping the hot reservoir temperature constant
- by increasing the temperature of the hot reservoir from 300°C to 350°C by keeping the cold reservoir temperature constant.
Which is the suitable method?
10 One mole of a van der Waals' gas obeying the equation `("P" + "a"/"V"^2)`(V - b) = RT undergoes the quasi-static cyclic process which is shown in the P-V diagram. The net heat absorbed by the gas in this process is ______
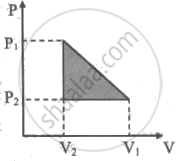
Draw a neat labelled P - V diagram for a typical heat engine.
