Advertisements
Advertisements
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Concept: Important Terms Pertaining to Coordination Compounds
Complete the following reactions
NH3+3Cl2(excess) ---->
Concept: Important Terms Pertaining to Coordination Compounds
What type of colloid is formed when a gas is dispersed in a liquid? Give an example
Concept: Properties of Colloidal Dispersions
Using IUPAC norms write the formulae for Dichloridobis(ethane-1,2-diamine)cobalt(III)
Concept: Importance and Applications of Coordination Compounds
What type of isomerism is shown by the complex [Co(NH3)5(SCN)]2+?
Concept: Isomerism in Coordination Compounds >> Structural Isomerism
Why is [NiCl4]2− paramagnetic while [Ni(CN)4]2− is diamagnetic? (Atomic number of Ni = 28)
Concept: Bonding in Coordination Compounds >> Magnetic Properties of Coordination Compounds
Why are low spin tetrahedral complexes rarely observed?
Concept: Bonding in Coordination Compounds >> Crystal Field Theory (CFT)
Write one difference in Multimolecular colloid and Associated colloid
Concept: Colloids
Write the structures of compounds A, B and C in the following reactions:

Concept: Important Terms Pertaining to Coordination Compounds
Write the structures of compounds A, B and C in the following reactions
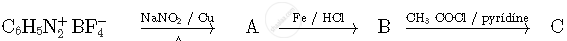
Concept: Important Terms Pertaining to Coordination Compounds
Physisorption is reversible while chemisorption is irreversible. Why ?
Concept: Types of Adsorption
Why is Butan-1-ol optically inactive but Butan-2-ol is optically active?
Concept: Isomerism in Coordination Compounds >> Stereoisomerism
Write the hybridization type and magnetic behaviour of the complex [Ni(CN)4]2−. (Atomic number of Ni = 28)
Concept: Bonding in Coordination Compounds >> Magnetic Properties of Coordination Compounds
Give reasons for the following observations :
NH3 gas adsorbs more readily than N2 gas on the surface of charcoal.
Concept: Adsorption >> Mechanism of Adsorption
Give reasons for the following observations :
Powdered substances are more effective adsorbents.
Concept: Types of Adsorption
Give one example each of 'oil in water' and 'water in oil' emulsion.
Concept: Emulsions
Give one example each of sol and gel.
Concept: Preparation of Colloids
Give one example each of sol and gel.
Concept: Preparation of Colloids
Define the following with a suitable example, of each:
Multimolecular colloid
Concept: Colloids
Define the following with a suitable example, of each:
Gel
Concept: Colloids
