Advertisements
Advertisements
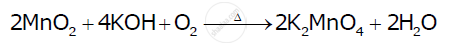
प्रश्न
Complete the following equations:

Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Give an example and suggest a reason for the following feature of the transition metal chemistry:
The highest oxidation state is exhibited in oxoanions of a metal.
Write the chemical equation for the preparation of 4 KMnO from 2. MnO Why does purple colour of acidified permanganate solution decolourise when it oxidises Fe2- to Fe3+ ?
On addition of small amount of \[\ce{KMnO4}\] to concentrated \[\ce{H2SO4}\], a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following.
Which of the following reactions are disproportionation reactions?
(a) \[\ce{Cu^{+} -> Cu^{2+} + Cu}\]
(b) \[\ce{3MnO^{-}4 + 4H^{+} -> 2MnO^{-}4 + MnO2 + 2H2O}\]
(c) \[\ce{2KMnO4 -> K2MnO4 + MnO2 + O2}\]
(d) \[\ce{2MnO^{-}4 + 3Mn^{2+} + 2H2O -> 5MnO2 + 4H^{+}}\]
Generally transition elements and their salts are coloured due to the presence of unpaired electrons in metal ions. Which of the following compounds are coloured?
(i) \[\ce{KMnO4}\]
(ii) \[\ce{Ce(SO4)2}\]
(iii) \[\ce{TiCl}\]
(iv) \[\ce{Cu2 Cl2}\]
When \[\ce{Cu^2+}\] ion is treated with \[\ce{KI}\], a white precipitate is formed. Explain the reaction with the help of chemical equation.
The oxidation state of manganese in the product obtained in a reaction of potassium permanganate and hydrogen peroxide in a basic medium is ______.
Complete the following ionic equation:
\[\ce{Cr2O^{2-}7 + 2OH^- ->}\]
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with SO2? Write the ionic equation for the reaction.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
