Advertisements
Advertisements
प्रश्न
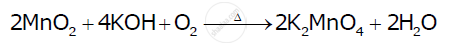
Complete the following equations:

Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Complete the following equations : 2 MnO2 + 4 KOH + O2 →
Name the oxometal anions of the first series of the transition metals in which the metal exhibits the oxidation state equal to its group number.
E0 value for Mn3+ Mn2+ couple is much more positive than that for Cr3-/ Cr2-.
Potassium permanganate acts as an oxidant in neutral, alkaline as well as acidic media. The final products obtained from it in the three conditions are, respectively.
What is the effect of pH on dichromate ion solution?
Potassium permanganate on heating at 513 K gives a product which is ______.
Complete the reaction mentioning all the products formed:
\[\ce{Cr2O^{2-}7 + 3H2S + 8H^+ ->}\]
Indicate the steps in the preparation of \[\ce{K2Cr2O2}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
