SSC (English Medium)
SSC (Marathi Semi-English)
Academic Year: 2024-2025
Date & Time: 3rd July 2025, 11:00 am
Duration: 2h
Advertisements
Note:
- All questions are compulsory.
- Use of a calculator is not allowed.
- The numbers to the right of the questions indicate full marks.
- In case of MCQs [Q. No. 1(A)] only the first attempt will be evaluated and will be given credit.
- Scientifically correct, labelled diagrams should be drawn wherever necessary.
- Each new question should be started on the new page.
Alkaline earth metals have valency 2. This means that their position in the modern periodic table is in ______.
Group 2
Group 16
Period 2
d-block
Chapter: [2] Periodic Classification of Elements
When the positive charge on an atom or an ion increases or the negative charge on them decreases it is called ______.
Reduction
Oxidation
Displacement
Decomposition
Chapter:
______ device is used to produce electricity.
Electric motor
Galvanometer
Electric generator
Voltmeter
Chapter:
Light changes its direction when going from one transparent medium to another transparent medium. This is called the ______of light.
Reflection
Dispersion
Refraction
Absorption
Chapter:
Height of the High earth orbit from the surface of the earth is ______.
= 20,000 km
= 27,000 km
< 30,780 km
> 35,780 km
Chapter:
Find the odd one out:
Fuse wire, bad conductor, rubber gloves, generator.
Chapter:
Complete the co-relation.
Red light wavelength close to : 700 nm : : Violet light wavelength close to : ______.
Chapter:
Match the pair:
| Group ‘A’ | Group ‘B’ | |
| Farsightedness | a. | Faraway object can be seen clearly |
| b. | Nearby object can be seen clearly | |
| c. | Faraway object cannot be seen clearly |
Chapter:
Write the electronic configuration of ‘Aluminium’.
Chapter:
State with reason whether the sentence is true or false.
The escape velocity on the moon is less than that on the earth.
Chapter: [10] Space Missions
Write scientific reason.
Sodium is always kept in kerosene.
Chapter:
Give scientific reason:
We cannot clearly see an object kept at a distance less than 25 cm from the eye.
Chapter:
Give a scientific reason.
Tungsten metal is used to make a solenoid type coil in an electric bulb.
Chapter: [4] Effects of Electric Current [14] The Electric Spark
Advertisements
An object takes 5 s to reach the ground from a height of 5 m on a planet. What is the value of g on the planet?
Chapter:
An element has its electron configuration as 2, 8, 2. Now answer the following question.
What is the atomic number of this element?
Chapter: [2] Periodic Classification of Elements
An element has its electron configuration as 2, 8, 2. Now answer the following question.
To which period does this element belong?
Chapter: [2] Periodic Classification of Elements
Complete the following reaction and give name of products:
CuSO4(aq) + Fe(s) → ______ + ______
Chapter:
SI units and CGS units of specific heat of an object.
Chapter:
Draw the electron dot diagram and structure of methane.
Chapter:
Write a short note.
Escape Velocity
Chapter: [1] Gravitation
State the modern periodic law.
Chapter: [2] Periodic Classification of Elements [11] School of Elements
How many rows and columns are there in the modern periodic table?
Chapter: [2] Periodic Classification of Elements [11] School of Elements
Answer the following sub-question regarding the modern periodic table:
What is the valency of halogens?
Chapter:
State whether the following reaction is endothermic or exothermic:
HCl + NaOH → NaCl + H2O + Heat
Chapter:
Advertisements
State whether the following reaction is endothermic or exothermic:
\[\ce{2KClO_{3(s)}->[\Delta] 2KCl_{(s)} + 3O2↑}\]
Chapter:
State whether the following reaction is endothermic or exothermic:
CaO + H2O → Ca(OH)2 + Heat
Chapter:
Identify the figure and give its use.

Chapter: [4] Effects of Electric Current [14] The Electric Spark
Identify the figure and give its use.

Chapter: [4] Effects of Electric Current [14] The Electric Spark
Identify the figure and give its use.

Chapter: [4] Effects of Electric Current
How much heat energy is necessary to raise the temperature of 5 kg of water from 20°C to 100°C?
Chapter: [5] Heat
Write the Cartesian sign convention for lenses.
Chapter:
Complete the following equations:
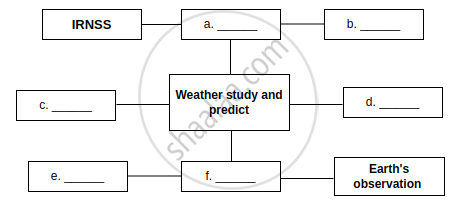
Chapter: [10] Space Missions
Write a balanced chemical equation for the thermit process reaction.
Chapter:
Give the chemical equation for the following reaction:
Burning of magnesium in air
Chapter:
Give the chemical equation for the following reaction:
Iron nail/dust is dropped in aqueous solution of copper sulphate.
Chapter:
Prove the statement.
A rainbow is the combined effect of the refraction, dispersion, and total internal reflection of light.
Chapter:
Complete the following table:
| Sr. No. | Straight chain of carbon atoms | Molecular formula | Name |
| 1. | C | CH4 | ______ |
| 2. | C-C | ______ | Ethane |
| 3. | C-C-C | C3H8 | ______ |
| 4. | C-C-C-C | ______ | Butane |
| 5. | C-C-C-C-C | C5H12 | ______ |
| 6. | C-C-C-C-C-C | ______ | Hexane |
| 7. | C-C-C-C-C-C-C | ______ | Heptane |
| 8. | C-C-C-C-C-C-C-C | C8H18 | ______ |
| 9. | C-C-C-C-C-C-C-C-C | ______ | Nonane |
| 10. | C-C-C-C-C-C-C-C-C-C | ______ | Decane |
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 10th Standard Science and Technology 1 with solutions 2024 - 2025
Previous year Question paper for Maharashtra State Board 10th Standard -2025 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science and Technology 1, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 10th Standard.
How Maharashtra State Board 10th Standard Question Paper solutions Help Students ?
• Question paper solutions for Science and Technology 1 will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
