(English Medium)
Academic Year: 2016-2017
Date & Time: 2nd April 2017, 1:00 pm
Duration: 2h
Advertisements
Question 1 is compulsory
Attempt any four questions from Question 2 to Question 7
The energy required to remove an electron from a neutral isolated gaseous atom and convert it into a positively charged gaseous ion is called ____________. (electron affinity, ionization potential,
electronegativity)
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
The compound that does not have a lone pair of electrons is __________. (water, ammonia, carbon tetrachloride)
Chapter:
When a metallic oxide is dissolved in water, the solution formed has a high concentration of ____________ ions (H+, h3O+, OH-)
Chapter: [4] Analytical Chemistry
Potassium sulfite on reacting with hydrochloric acid releases _______ gas. (Cl2, SO2, H2S)
Chapter: [8] Study of Compounds A. Hydrogen Chloride
The compound formed when ethene reacts with Hydrogen is ________. (CH4, C2H6, C3H8)
Chapter:
Choose the correct answer from the options given below:
A chloride which forms a precipitate that is soluble in excess of ammonium hydroxide is:
1) Calcium chloride
2) Ferrous chloride
3) Ferric chloride
4) Copper chloride
Chapter: [4] Analytical Chemistry
Choose the correct answer from the options given below:
If the molecular formula of an organic compound is C10H18 it is:
1) alkene
2) alkane
3) alkyne
4) Not a hydrocarbon
Chapter:
Choose the correct answer from the options given below:
Which of the following is a common characteristic of a covalent compound?
1) high melting point
2) consists of molecules
3) always soluble in water
4) Conducts electricity when it is in the molten state
Chapter: [2] Chemical Bonding
Choose the correct answer from the options given below
To increase the pH value of a neutral solution, we should add:
1) an acid
2) an acid salt
3) an alkali
4) a salt
Chapter:
Choose the correct answer from the options given below:
Anhydrous iron(III) chloride is prepared by:
1) direct combination
2) simple displacement
3) decomposition
4) neutralization
Chapter:
Identify the substance underlined, in the following case:
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide.
Chapter: [4] Analytical Chemistry
Identify the substance underlined, in the following case
The electrolyte used for electroplating an article with silver.
Chapter: [6] Electrolysis
Identify the substance underlined, in the following case
The particles present in a liquid such as kerosene, that is a nonelectrolyte.
Chapter:
Identify the substance underlined, in the following case
An organic compound containing – COOH functional group
Chapter: [12.1] Organic Compounds
Identify the substance underlined, in the following case
A solid formed by reaction of two gases, one of which is acidic and the other basic in nature
Chapter: [3.2] Salts and their Preparations
Write a balanced chemical equation for the following:
Action of cold and dilute Nitric acid on Copper
Chapter: [10] Study of Compounds C. Nitric Acid
Write a balanced chemical equation for the following:
Reaction of Ammonia with heated copper oxide.
Chapter: [9] Study of Compounds B. Ammonia
Write a balanced chemical equation for the following:
Preparation of methane from iodomethane.
Chapter: [3.2] Salts and their Preparations
Write a balanced chemical equation for the following:
Action of concentrated sulphuric acid on Sulphur.
Chapter: [11] Study of Compounds D. Sulphuric Acid
Write a balanced chemical equation for the following:
Laboratory preparation of ammonia from ammonium chloride.
Chapter:
State one relevant observation for given reactions:
Addition of ethyl alcohol to acetic acid in the presence of concentrated Sulphuric acid
Chapter: [11] Study of Compounds D. Sulphuric Acid
State one relevant observation for given reactions:
Action of dilute Hydrochloric acid on iron (II) sulfide.
Chapter: [8] Study of Compounds A. Hydrogen Chloride
State one relevant observation for given reactions:
Action of Sodium hydroxide solution on ferrous sulfate solution.
Chapter: [4] Analytical Chemistry
State one relevant observation for given reactions:
Burning of ammonia in air.
Chapter: [9] Study of Compounds B. Ammonia
State one relevant observation for given reactions:
Action of concentrated Sulphuric acid on hydrated copper sulfate
Chapter: [11] Study of Compounds D. Sulphuric Acid
Draw the structural formula for 2, 3-dimethylbutane.
Chapter:
Draw the structural formula for Diethyl ether
Chapter: [12.1] Organic Compounds
Select the process from the following which matches the given description.
(calcination, roasting, pulverisation, smelting)
Crushing of the ore into a fine powder.
Chapter:
Select the process from the following which matches the given description.
(calcination, roasting, pulverisation, smelting)
Heating of the ore in the absence of air to a high temperature.
Chapter:
Calculate the number of gram atoms in 4.6 grams of sodium (Na = 23)
Chapter:
Calculate the percentage of water of crystallization in CuSO4.5H2O
(H = 1, O = 16, S = 32, Cu = 64)
Chapter: [5.2] Relative Atomic Mass, Relative Molecular Mass and Mole Concept
A compound of X and Y has the empirical formula XY2. Its vapor density is equal to its empirical formula weight. Determine its molecular formula.
Chapter: [5.3] Percentage Composition, Empirical and Molecular Formulae
Match the atomic number 2, 4, 8, 15 and 19 with each of the following
1) A solid non metal belonging to the third period.
2) A metal of valency 1.
3) A gaseous element with valency 2.
4) An element belonging to Group 2
5) A rare gas.
Chapter:
Arrange the following as per the instruction given in the brackets:
He, Ar, Ne (Increasing order of the number of electron shells)
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
Arrange the following as per the instruction given in the brackets:
Na, Li, K (Increasing Ionisation Energy)
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
Arrange the following as per the instruction given in the brackets :
F, Cl, Br (Increasing electronegativity)
Chapter:
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
Advertisements
State the type of bonding in the following molecule.
Water
Chapter: [2] Chemical Bonding
State the type of Bonding in the following molecule:
Calcium oxide
Chapter:
How will you distinguish between Ammonium hydroxide and Sodium hydroxide using copper sulphate solution?
Chapter: [4] Analytical Chemistry
How will you distinguish between dilute hydrochloric acid and dilute sulphuric acid using lead nitrate solution?
Chapter: [11] Study of Compounds D. Sulphuric Acid
Identify the salts P and Q from the observations given below:
On performing the flame test salt P produces a lilac coloured flame and its solution gives a white precipitate with silver nitrate solution, which is soluble in Ammonium hydroxide solution.
Chapter: [4] Analytical Chemistry
When dilute HCl is added to a salt Q, a brisk effervescence is produced and the gas turns lime water milky.
When NH4OH solution is added to the above mixture (after adding dilute HCl), it produces a white precipitate which is soluble in excess NH4OH solution.
Chapter: [8] Study of Compounds A. Hydrogen Chloride
Draw an electron dot diagram to show the formation of each of the following compounds:
Methane
[H = 1, C = 6, Mg = 12, Cl = 17]
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
Draw an electron dot diagram to show the formation of each of the following compounds:
Magnesium Chloride
[H = 1, C = 6, Mg = 12, Cl = 17]
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
State the observations at the anode and at the cathode during the electrolysis of fused lead bromide using graphite electrodes
Chapter:
State the observations at the anode and at the cathode during the electrolysis of copper sulphate solution using copper electrodes.
Chapter:
Select the ion in the following case, that would get selectively discharged from the aqueous mixture of the ions listed below:
\[\ce{SO^2-_4, NO^-_3 {and} OH-}\]
Chapter: [6] Electrolysis
Select the ion in the following case, that would get selectively discharged from the aqueous mixture of the ions listed below:
Pb2+, Ag+ and Cu2+
Chapter: [6] Electrolysis
Certain blank spaces are left in the following table and these are labelled as A, B, C, D
and E. Identify each of them
| Lab preparation of | Reactants used | Products formed | Drying Agent | Method of collection |
|
| 1 | HCl gas | NaCl + H2SO4 |  |
conc. H2SO4 | 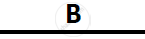 |
| 2 | NH3 gas |
|
Mg(OH)2 NH3 |
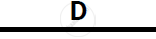 |
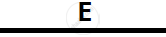 |
Chapter:
Write balanced chemical equations to show : The oxidizing action of conc. Sulphuric acid on carbon
Chapter: [11] Study of Compounds D. Sulphuric Acid
Write balanced chemical equations to show The behavior of H2SO4 as an acid when it reacts with Magnesium
Chapter: [8] Study of Compounds A. Hydrogen Chloride
Write balanced chemical equations to show The dehydrating property of conc. Sulphuric acid with sugar.
Chapter: [11] Study of Compounds D. Sulphuric Acid
Write balanced chemical equations to show how SO3 is converted to Sulphuric acid in the contact process.
Chapter: [11] Study of Compounds D. Sulphuric Acid
Advertisements
Propane burns in air according to the following equation:
C3H8 + 5O2 → 3CO2 + 4H2O
What volume of propane is consumed on using 1000 cm3 of air, considering only 20% of air contains oxygen?
Chapter: [5.1] Gay-Lussac's Law and Avogadro's Law
The mass of 11.2 litres of a certain gas at s.t.p. is 24 g. Find the gram molecular mass of the gas
Chapter:
A gas cylinder can hold 1 kg of hydrogen at room temperature and pressure
1) Find the number of moles of hydrogen present.
2) What weight of CO2 can the cylinder hold under similar conditions of temperature and pressure? (H = 1, C = 12, O = 16)
3) If the number of molecules of hydrogen in the cylinder is X, calculate the number of CO2 molecules in the cylinder under the same conditions of temperature and pressure.
4) State the law that helped you to arrive at the above result.
Chapter: [5.2] Relative Atomic Mass, Relative Molecular Mass and Mole Concept
Write a balanced chemical equation for the preparation of the following salts
Copper carbonate
Chapter: [3.2] Salts and their Preparations
Write a balanced chemical equation for the preparation of the following salts
Ammonium sulphate crystals
Chapter:
Give a balanced chemical equation for Action of conc. Nitric acid on Sulphur
Chapter: [10] Study of Compounds C. Nitric Acid
Give a balanced chemical equation for Catalytic oxidation of Ammonia
Chapter: [9] Study of Compounds B. Ammonia
Give a balanced chemical equation for Laboratory preparation of Nitric acid.
Chapter: [10] Study of Compounds C. Nitric Acid
Give a balanced chemical equation for Reaction of Ammonia with Nitric acid
Chapter: [9] Study of Compounds B. Ammonia
Identify the term or substance based on the descriptions given below
Ice like crystals formed on cooling an organic acid sufficiently.
Chapter: [12.6] Carboxylic Acids
Identify the term or substance based on the descriptions given below:
Hydrocarbon containing a triple bond used for welding purposes.
Chapter:
Identify the term or substance based on the descriptions given below:
The property by virtue of which the compound has the same molecular formula but different structural formulae.
Chapter: [5.3] Percentage Composition, Empirical and Molecular Formulae
The compound formed where two alkyl groups are linked by  group.
group.
Chapter:
Give a balanced chemical equation for Preparation of ethane from Sodium propionate
Chapter:
Give a balanced chemical equation for Action of alcoholic KOH on bromethane
Chapter:
Name the following :
The process of coating of iron with zinc
Chapter: [6] Electrolysis
Name the following:
An alloy of lead and tin that is used in electrical circuits.
Chapter:
Name the following:
An ore of zinc containing its sulphide.
Chapter:
Name the following:
A metal oxide that can be reduced by hydrogen.
Chapter: [7.2] Stages Involved in the Extraction of Metals
Answer the following questions with respect to the electrolytic process in the extraction of aluminum:
Identify the components of the electrolyte other than pure alumina and the role played by each
Chapter:
Answer the following questions with respect to the electrolytic process in the extraction of aluminum:
Explain why powdered coke is sprinkled over the electrolytic mixture.
Chapter:
Complete the following by selecting the correct option from the choices given:
The metal which does not react with water or dilute H2SO4 but reacts with concentrated H2SO4 is _________. (Al/Cu/Zn/Fe)
Chapter: [8] Study of Compounds A. Hydrogen Chloride
Complete the following by selecting the correct option from the choices given:
The metal whose oxide, which is amphoteric, is reduced to metal by carbon reduction ____________. (Fe/Mg/Pb/Al)
Chapter: [7.2] Stages Involved in the Extraction of Metals
The divalent metal whose oxide is reduced to metal by electrolysis of its fused salt is ______.
Al
Na
Mg
K
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CISCE previous year question papers ICSE Class 10 Chemistry with solutions 2016 - 2017
Previous year Question paper for CISCE ICSE Class 10 -2017 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CISCE ICSE Class 10 .
How CISCE ICSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
