Advertisements
Advertisements
प्रश्न
When dilute HCl is added to a salt Q, a brisk effervescence is produced and the gas turns lime water milky.
When NH4OH solution is added to the above mixture (after adding dilute HCl), it produces a white precipitate which is soluble in excess NH4OH solution.
Advertisements
उत्तर
ZnCO3
APPEARS IN
संबंधित प्रश्न
Name the drying agent used to dry HCl gas.
How can you prove that hydrochloric acid contain :
Chlorine?
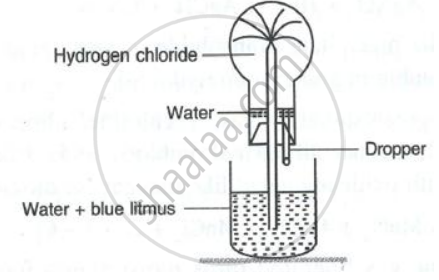
Hydrogen chloride gas is very soluble in water. It is dissolved in water to prepare hydrochloric acid by using an anti-suction device, as shown in the diagram. Very briefly explain how this device prevents the suction of water into the flask, in which hydrogen chloride gas is produced.
Give reason for the following:
Dilute hydrochloric acid cannot be concentrated by distilling (boiling) the dilute acid.
Dilute hydrochloric acid is added in turn to a mixture of iron and sulphur and to the compound formed between iron and sulphur. Name the gas formed in each case.
Hydrogen chloride dissolves in water to form an acidic solution.
Name the experiment which demonstrates that hydrogen chloride is very soluble in water.

(a) Name the experiment illustrate above.
(b) Which property of hydrogen chloride is demonstrated by this experiment?
(c) State the colour of the water that has entered the round-bottomed flask.
The following question is pertaining to the laboratory preparation of hydrogen chloride gas.
Write the equation for its preparation, mentioning the condition required.
- Name the experiment illustrated below.
- State the colour of the water that has entered the round-bottomed flask.
Identify the terms for the following:
A method used to collect HCl gas.
