Advertisements
Advertisements
प्रश्न
Draw an electron dot diagram to show the formation of each of the following compounds:
Magnesium Chloride
[H = 1, C = 6, Mg = 12, Cl = 17]
Advertisements
उत्तर
Magnesium atom loses 2 electrons to attain a stable electronic configuration and becomes a cation.
Example:
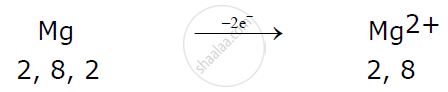
A non-metallic atom like chlorine gains 1 electron to attain a stable electronic configuration and becomes an anion.
Example:
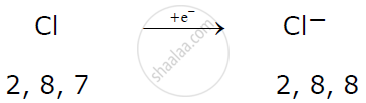
Cations and anions are oppositely charged particles which attract one another to form an electrovalent bond leading to the formation of an electrovalent compound
Here magnesium donates one electron each with two chlorine atoms resulting in the formation of magnesium chloride.

APPEARS IN
संबंधित प्रश्न
Draw an electron dot diagram to show the formation of each of the following compounds:
Methane
[H = 1, C = 6, Mg = 12, Cl = 17]
Give reason:
Electronegativity of chorine is higher than Sulphur?
Explain the following:
The reducing power of element increases down in the group while decreases in a period.
Which has higher E.A. fluorine or Neon?
A, B, C are three elements in which B is an inert gas other than helium.With this information complete the following table.
| Element | Atomic number | No. of electrons in the valence shell | Group to which the element belongs |
| A | Z - 1 | ||
| B | Z | ||
| C | Z + 1 |
Also, explain the following : Electron affinity of B is Zero.
State whether the following statement is true or false
Al2O3 is an amphoteric oxide.
State whether the following statement is true or false
The elements with higher electron affinity have higher ionization potential.
What is electron affinity?
Neon shows zero electron affinity due to ______.
Arrange the following as per instruction given in the bracket.
Cl, F, Br, I (increasing electron affinity)
