Advertisements
Advertisements
प्रश्न
Draw an electron dot diagram to show the formation of each of the following compounds:
Magnesium Chloride
[H = 1, C = 6, Mg = 12, Cl = 17]
Advertisements
उत्तर
Magnesium atom loses 2 electrons to attain a stable electronic configuration and becomes a cation.
Example:
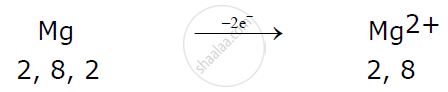
A non-metallic atom like chlorine gains 1 electron to attain a stable electronic configuration and becomes an anion.
Example:
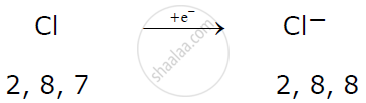
Cations and anions are oppositely charged particles which attract one another to form an electrovalent bond leading to the formation of an electrovalent compound
Here magnesium donates one electron each with two chlorine atoms resulting in the formation of magnesium chloride.

APPEARS IN
संबंधित प्रश्न
State the factors on which electron affinity depends ?
Give reason:
Electron affinity of halogens is comparatively high ?
Give reason:
Electronegativity of chorine is higher than Sulphur?
Explain the following:
The reducing power of element increases down in the group while decreases in a period.
Which has higher E.A. fluorine or Neon?
Choose the correct answer:
Among the period 2 elements the one which has high electron affinity is
In a period, increase in electron affinity increases ______.
Neon shows zero electron affinity due to ______.
Among period 2 elements A, B, C and D, the one which has highest electron affinity is ______.
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element A would probably have ______ electron affinity than B.
