Advertisements
Advertisements
प्रश्न
Which of the following graphs represents exothermic reaction?
(a)

(b)
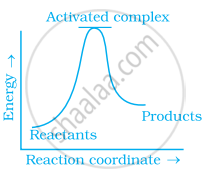
(c)
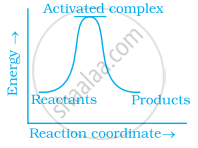
पर्याय
(a) only
(b) only
(c) only
(a) and (b)
Advertisements
उत्तर
(a) only
Explanation:
Exothermic reaction is a chemical reaction in which the enthalpy change is negative and energy is released in the form of light and heat.
As we all know, ΔH is negative.
Activation energy (forward reaction) – (backward reaction)
(Forward reaction) – Ea (backward reaction)
The product's exothermic energy exceeds the activation energy of the reactant.
APPEARS IN
संबंधित प्रश्न
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
The rate of the chemical reaction doubles for an increase of 10 K in absolute temperature from 298 K. Calculate Ea.
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/T` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
Consider a certain reaction \[\ce{A -> Products}\] with k = 2.0 × 10−2 s−1. Calculate the concentration of A remaining after 100 s if the initial concentration of A is 1.0 mol L−1.
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
Calculate activation energy for a reaction of which rate constant becomes four times when temperature changes from 30 °C to 50 °C. (Given R = 8.314 JK−1 mol−1).
A first-order reaction is 50% completed in 40 minutes at 300 K and in 20 minutes at 320 K. Calculate the activation energy of the reaction. (Given : log 2 = 0·3010, log 4 = 0·6021, R = 8·314 JK–1 mol–1)
Explain the following terms :
Half life period of a reaction (t1/2)
Write a condition under which a bimolecular reaction is kinetically first order. Give an example of such a reaction. (Given : log2 = 0.3010,log 3 = 0.4771, log5 = 0.6990).
The chemical reaction in which reactants require high amount of activation energy are generally ____________.
Activation energy of a chemical reaction can be determined by ______.
Which of the following statements are in accordance with the Arrhenius equation?
(i) Rate of a reaction increases with increase in temperature.
(ii) Rate of a reaction increases with decrease in activation energy.
(iii) Rate constant decreases exponentially with increase in temperature.
(iv) Rate of reaction decreases with decrease in activation energy.
Why does the rate of a reaction increase with rise in temperature?
Thermodynamic feasibility of the reaction alone cannot decide the rate of the reaction. Explain with the help of one example.
The slope of Arrhenius Plot `("In" "k" "v"//"s" 1/"T")` of first-order reaction is −5 × 103 K. The value of Ea of the reaction is. Choose the correct option for your answer. [Given R = 8.314 JK−1mol−1]
It is generally observed that the rate of a chemical reaction becomes double with every 10°C rise in temperature. If the generalisation holds true for a reaction in the temperature range of 298 K to 308 K, what would be the value of activation energy (Ea) for the reaction?
Assertion (A): A reaction can have zero activation energy.
Reason (R): The minimum extra amount of energy absorbed by reactant molecules so that their energy becomes equal to the threshold value is called activation energy.
In the light of the above statements, choose the correct answer from the options given below:
Given below are two statements:
Statement I: The nutrient deficient water bodies lead to eutrophication.
Statement II: Eutrophication leads to decrease in the level of oxygen in the water bodies.
In the light of the above statements, choose the correct answer from the options given below:
