Advertisements
Advertisements
प्रश्न
The rate of the chemical reaction doubles for an increase of 10 K in absolute temperature from 298 K. Calculate Ea.
Advertisements
उत्तर
Given: T1 = 298 K
T2 = 308 K
`K_2/k_1` = 2
R = 8.314 JK−1 mol−1
Ea = ?
According to the Arrhenius equation,
`log k_2/k_1 = (E_a)/(2.303 R) [1/T_1 - 1/T_2]`
∴ log 2 = `E_a/(2.303 xx 8.314) [1/298 - 1/308]`
0.3010 = `E_a/(2.303 xx 8.314)^-1 xx 10/(298 xx 308)`
∴ Ea = `(0.3010 xx 2.303 xx 8.314 xx 298 xx 308)/10`
= `528977.78/10`
= 52897.7 J mol−1
= 52.897 kJ mol−1
APPEARS IN
संबंधित प्रश्न
(b) Rate constant ‘k’ of a reaction varies with temperature ‘T’ according to the equation:
`logk=logA-E_a/2.303R(1/T)`
Where Ea is the activation energy. When a graph is plotted for `logk Vs. 1/T` a straight line with a slope of −4250 K is obtained. Calculate ‘Ea’ for the reaction.(R = 8.314 JK−1 mol−1)
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/T` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
The rate constant of a first order reaction are 0.58 S-1 at 313 K and 0.045 S-1 at 293 K. What is the energy of activation for the reaction?
What is the effect of adding a catalyst on Activation energy (Ea)
Write a condition under which a bimolecular reaction is kinetically first order. Give an example of such a reaction. (Given : log2 = 0.3010,log 3 = 0.4771, log5 = 0.6990).
Predict the main product of the following reactions: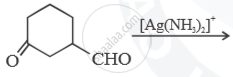
Which of the following graphs represents exothermic reaction?
(a)

(b)
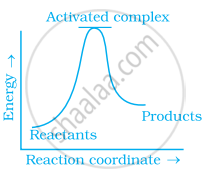
(c)
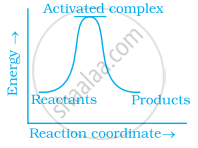
During decomposition of an activated complex:
(i) energy is always released
(ii) energy is always absorbed
(iii) energy does not change
(iv) reactants may be formed
Which of the following statements are in accordance with the Arrhenius equation?
(i) Rate of a reaction increases with increase in temperature.
(ii) Rate of a reaction increases with decrease in activation energy.
(iii) Rate constant decreases exponentially with increase in temperature.
(iv) Rate of reaction decreases with decrease in activation energy.
Mark the incorrect statements:
(i) Catalyst provides an alternative pathway to reaction mechanism.
(ii) Catalyst raises the activation energy.
(iii) Catalyst lowers the activation energy.
(iv) Catalyst alters enthalpy change of the reaction.
The reaction between \[\ce{H2(g)}\] and \[\ce{O2(g)}\] is highly feasible yet allowing the gases to stand at room temperature in the same vessel does not lead to the formation of water. Explain.
Total number of vibrational degrees of freedom present in CO2 molecule is
The rate constant for a reaction is 1.5 × 10–7 sec–1 at 50°C. What is the value of activation energy?
An exothermic reaction X → Y has an activation energy 30 kJ mol-1. If energy change ΔE during the reaction is - 20 kJ, then the activation energy for the reverse reaction in kJ is ______.
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below

The reaction must be:
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
It is generally observed that the rate of a chemical reaction becomes double with every 10°C rise in temperature. If the generalisation holds true for a reaction in the temperature range of 298 K to 308 K, what would be the value of activation energy (Ea) for the reaction?
