Advertisements
Advertisements
प्रश्न
What is the rate constant?
What do you understand by rate constant?
What is meant by the rate constant of a reaction?
Advertisements
उत्तर १
When the concentration (active mass) of each reactant is at unity, the rate constant of a chemical reaction equals the rate of the reaction.
∵ Rate of reaction ∝ [Reactant]
If A → product, then
rate (R) = k[A]
If [A] = 1, then rate, R = k,
where k is the rate constant.
उत्तर २
- The rate constant of a reaction is equal to the rate of reaction when the concentration of each of the reactants is unity.
- At a specific temperature, a reaction's rate constant has a fixed value. As the temperature rises, so does its worth. It is independent of the reactant concentrations at the initiation.
APPEARS IN
संबंधित प्रश्न
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume.
\[\ce{SO2Cl2_{(g)} -> SO2_{(g)} + Cl2_{(g)}}\]
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.5 |
| 2 | 100 | 0.6 |
Calculate the rate of the reaction when total pressure is 0.65 atm.
The rate constant for a first order reaction is 60 s−1. How much time will it take to reduce the initial concentration of the reactant to its `1/16`th value?
For the decomposition of azoisopropane to hexane and nitrogen at 543 K, the following data are obtained.
| t (sec) | P(mm of Hg) |
| 0 | 35.0 |
| 360 | 54.0 |
| 720 | 63.0 |
Calculate the rate constant.
Define order of reaction. How does order of a reaction differ from molecularity for a complex reaction?
The reaction X → product
Follow first order of kinetics. In 40 minutes the concentration of 'X' changes from 0.1 m to 0.025. M. The rate of reaction when concentration of X is 0.01 m is.
Observe the graph shown in figure and answer the following questions:
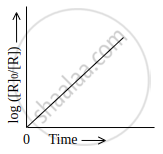
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
The reaction \[\ce{SO2Cl2(g) -> SO2(g) + Cl2(g)}\] is a first-order gas reaction with k = 2.2 × 10−5 sec−1 at 320°C. The percentage of SO2Cl2 is decomposed on heating this gas for 90 min, is ______%.
The slope in the plot of ln[R] vs. time for a first order reaction is ______.
Slove: \[\ce{2NOBr -> 2NO_{2(g)} + Br_{2(g)}}\]
For the above reaction, the rate law is rate = k[NOBr]2. If the rate of reaction is 6.5 × 10−6 mol L−1 s−1 at 2 × 10−3 mol L−1 concentration of NOBr, calculate the rate constant k for the reaction.
The rate constant for the reaction:
\[\ce{2N2O_{(s)} ->2N2O4_{(g)}}\] is 4.98 × 10-4 s-1.
The order of the reaction is ______.
