Advertisements
Advertisements
प्रश्न
How can you remove the hard calcium carbonate layer of the egg without damaging its semiprermiable membrane? Can this egg be inserted into a bottle with a narrow neck without distorting its shape? Explain the process involved.
Advertisements
उत्तर
(i) When egg is placed in dilute mineral acid solution (preferably dilute \[\ce{HCl}\] solution), the hard external \[\ce{CaCO3}\] layer of the egg dissolves out/removed without damaging its semipermeable menbrane.
(ii) Yes, this egg can be inserted into a bottle with a narrow neck without distorting is shape.
This process invloved utilising phenomenon of osmosis is explained as below:
Egg is placed in mineral acid solution - after time egg is removed and placed in a hypertonic solution* - Size of the egg gradually decreases after some time and it shrivles due to osmasis. SInce the egg has shrivelled it can, now be inserted easily into a bottle with narrow mouth. The egg is, therefore, placed in a battle with narrow neck and then a hypotonic solution is filled into this bottle. On adding hypotonic solution**, egg regains shape due to osmosis.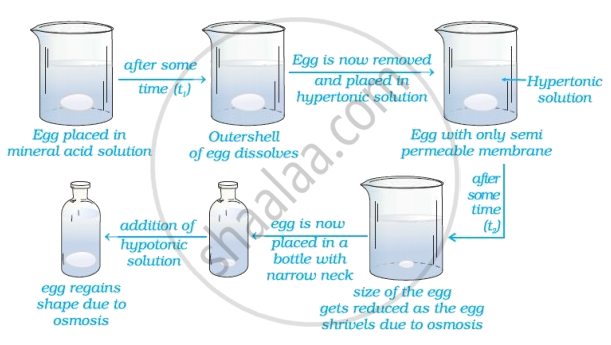
• Hypertonic solution - is a solution with higher salt concentration than that of the normal body cells so that the solvent/water is drawn out of the cell by osmosis; or any solution with higher osmotic pressure than another solution is called "Hypertonic solution".
• Hypotonic solution is a solution with lower salt concentration than that of the normal body cells so that water/solvent flows into the cell by osmosis; or - hypotonic solution is a solution which has lower osmotic pressure than the other solution.
APPEARS IN
संबंधित प्रश्न
At 300 K, 36 g of glucose present in a litre of its solution has an osmotic pressure of 4.98 bar. If the osmotic pressure of the solution is 1.52 bars at the same temperature, what would be its concentration?
Which of the following 0.1 M aqueous solutions will exert the highest osmotic pressure?
Answer the following in one or two sentences.
A solution concentration is expressed in molarity and not in molality while considering osmotic pressure. Why?
Answer the following.
A solvent and its solution containing a nonvolatile solute are separated by a semipermeable membrane. Does the flow of solvent occur in both directions? Comment giving a reason.
What are hypertonic solutions?
Define Osmosis.
Two solutions have different osmotic pressures. The solution of higher osmotic pressure is called ____________.
Osmotic pressure of a solution is 0.0821 atm at a temperature of 300 K. The concentration in moles/litre will be:
Which of the following statements is false?
Give an example of a material used for making semipermeable membrane for carrying out reverse osmosis.
Discuss biological and industrial importance of osmosis.
Isotonic solutions have same
Which one of the following is a colligative property?
Name the four colligative properties that are oftently used for determination of molecular mass.
Write the condition of reverse osmosis.
Calculate the osmotic pressure of 0.03 mole of non electrolyte solute dissolved in 0.1 dm3 of water at 300 K. [R = 0.082 dm3 atm mol-1 K-1]
The 'X' g nonvolatile solute having molar mass 196 g mol-1 is dissolved in 3 dm³ water. Calculate the value of 'X' if resulting solution has osmotic pressure 0.8 atm at 300 К.
(R = 0.0821dm³ atm K-1 mol-1)
The plot of osmotic pressure (π) vs concentration (mol L−1) for a solution gives a straight line with slope 25.73 L bar mol−1. The temperature at which the osmotic pressure measurement is done is ______.
(Use R = 0.083 L bar mol−1 K−1)
Calculate the osmotic pressure of 0.2 M aqueous solution of nonelectrolyte at 300 K.
[R = 0.082 atm dm3 mol−1K−1]
