Advertisements
Advertisements
प्रश्न
For the given cell, \[\ce{Mg | Mg^{2+} || Cu^{2+} | Cu}\]
(i) \[\ce{Mg}\] is cathode
(ii) \[\ce{Cu}\] is cathode
(iii) The cell reaction is \[\ce{Mg^+ Cu^{2+} -> Mg^{2+} + Cu}\]
(iv) \[\ce{Cu}\] is the oxidising agent
Advertisements
उत्तर
(ii) \[\ce{Cu}\] is cathode
(iii) The cell reaction is \[\ce{Mg^+ Cu^{2+} -> Mg^{2+} + Cu}\]
Explanation:
(i) Left side of cell reaction represents oxidation half-cell i.e., oxidation of \[\ce{Mg}\] and right side of cell represents reduction half-cell reactions i.e., reduction of copper.
(ii) \[\ce{Cu}\] is reduced and reduction occurs at cathode.
(iii) \[\ce{Mg}\] is oxidized and oxidation occurs at anode.
(iv) Whole cell reaction can be written as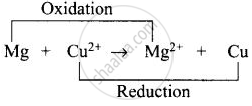
APPEARS IN
संबंधित प्रश्न
Construct a labelled diagram for the following cell:
`Zn|Zn^(2+)(1M)||H^+(1M)|H_(2(g,1atm))|Pt`
Among Zn and Cu, which would occur more readily in nature as metal and which as an ion?
What is the SI unit tor electrochemical equivalent?
If one mole electrons is passed through the solutions of CrCl3, AgNO3 and NiSO4, in what ratio Cr, Ag and Ni will be deposited at the electrodes?
In the electrochemical cell: Zn|ZnSO4 (0.01 M)||CuSO4 (1.0 M)|Cu, the emf of this Daniel cell is E1. When the concentration of ZnSO4 is changed to 1.0 M and that CuSO4 changed to 0.01 M, the emf changes to E2. From the above, which one is the relationship between E1 and E2?
Describe the electrolysis of molten NaCl using inert electrodes.
Two metals M1 and M2 have reduction potential values of −xV and +yV respectively. Which will liberate H2 and H2SO4.
Which of the following statement is correct?
Use the data given in below find out the most stable ion in its reduced form.
`"E"_("Cr"_2"O"_7^(2-)//"Cr"^(3+))^⊖`= 1.33 V `"E"_("Cl"_2//"Cl"^-)^⊖` = 1.36 V
`"E"_("MnO"_4^-//"Mn"^(2+))^⊖` = 1.51 V `"E"_("Cr"^(3+)//"Cr")^⊖` = - 0.74 V
Consider the following diagram in which an electrochemical cell is coupled to an electrolytic cell. What will be the polarity of electrodes ‘A’ and ‘B’ in the electrolytic cell?
Match the terms given in Column I with the units given in Column II.
| Column I | Column II |
| (i) Λm | (a) S cm-¹ |
| (ii) ECell | (b) m-¹ |
| (iii) K | (c) S cm2 mol-¹ |
| (iv) G* | (d) V |
Assertion: ECell should have a positive value for the cell to function.
Reason: `"E"_("cathode") < "E"_("anode")`
The electrochemical cell stops working after some time because
Given the data at 25°C
\[\ce{Ag + I- -> AgI + e-}\]; E° = – 0.152 V
\[\ce{Ag -> Ag+ + e-}\]; E° = – 0.800 V
The value of log Ksp for AgI is ______.
Cell reaction is spontaneous when
If the half-cell reaction A + e– → A– has a large negative reduction potential, it follow that:-
In a solution of CuSO4, how much time will be required to precipitate 2 g copper by 0.5 ampere current?
Galvanic cell is a device in which ______.
State the term for the following:
Two metal plates or wires through which the current enters and leaves the electrolytic cell.
