Advertisements
Advertisements
प्रश्न
For the given cell, \[\ce{Mg | Mg^{2+} || Cu^{2+} | Cu}\]
(i) \[\ce{Mg}\] is cathode
(ii) \[\ce{Cu}\] is cathode
(iii) The cell reaction is \[\ce{Mg^+ Cu^{2+} -> Mg^{2+} + Cu}\]
(iv) \[\ce{Cu}\] is the oxidising agent
Advertisements
उत्तर
(ii) \[\ce{Cu}\] is cathode
(iii) The cell reaction is \[\ce{Mg^+ Cu^{2+} -> Mg^{2+} + Cu}\]
Explanation:
(i) Left side of cell reaction represents oxidation half-cell i.e., oxidation of \[\ce{Mg}\] and right side of cell represents reduction half-cell reactions i.e., reduction of copper.
(ii) \[\ce{Cu}\] is reduced and reduction occurs at cathode.
(iii) \[\ce{Mg}\] is oxidized and oxidation occurs at anode.
(iv) Whole cell reaction can be written as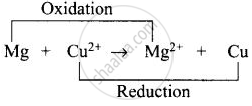
APPEARS IN
संबंधित प्रश्न
Arrange the following metals in the order in which they displace each other from the solution of their salts.
Al, Cu, Fe, Mg and Zn.
E°cell for the given redox reaction is 2.71 V
Mg(s) + Cu2+ (0.01 M) → Mg2+ (0.001 M) + Cu(s)
Calculate Ecell for the reaction. Write the direction of flow of current when an external opposite potential applied is
(i) less than 2.71 V and
(ii) greater than 2.71 V
If 'I' stands for the distance between the electrodes and 'a' stands for the area of cross-section of the electrode, `"l"/"a"` refers to ____________.
At 25°C, the emf of the following electrochemical cell.
\[\ce{Ag_{(s)} | Ag^+ (0.01 M) | | Zn^{2+} {(0.1 M)} | Zn_{(s)}}\] will be:
(Given \[\ce{E^0_{cell}}\] = −1.562 V)
In the electrochemical cell: Zn|ZnSO4 (0.01 M)||CuSO4 (1.0 M)|Cu, the emf of this Daniel cell is E1. When the concentration of ZnSO4 is changed to 1.0 M and that CuSO4 changed to 0.01 M, the emf changes to E2. From the above, which one is the relationship between E1 and E2?
A certain current liberated 0.504 gm of hydrogen in 2 hours. How many grams of copper can be liberated by the same current flowing for the same time through copper sulphate solution.
Describe the construction of Daniel cell. Write the cell reaction.
Can Fe3+ oxidises bromide to bromine under standard conditions?
Given: \[\ce{E^0_{{Fe^{3+}|Fe^{2+}}}}\] = 0.771 V
\[\ce{E^0_{{Br_{2}|Br^-}}}\] = −1.09 V
A copper electrode is dipped in 0.1 M copper sulphate solution at 25°C. Calculate the electrode potential of copper.
[Given: \[\ce{E^0_{{Cu^{2+}|Cu}}}\] = 0.34 V]
Which of the following statement is not correct about an inert electrode in a cell?
Use the data given in below find out which option the order of reducing power is correct.
`"E"_("Cr"_2"O"_7^(2-)//"Cr"^(3+))^⊖`= 1.33 V `"E"_("Cl"_2//"Cl"^-)^⊖` = 1.36 V
`"E"_("MnO"_4^-//"Mn"^(2+))^⊖` = 1.51 V `"E"_("Cr"^(3+)//"Cr")^⊖` = - 0.74 V
A galvanic cell has electrical potential of 1.1V. If an opposing potential of 1.1V is applied to this cell, what will happen to the cell reaction and current flowing through the cell?
Consider a cell given below:
\[\ce{Cu | Cu^{2+} || Cl^{-} | Cl_{2},Pt}\]
Write the reactions that occur at anode and cathode
Given the data at 25°C
\[\ce{Ag + I- -> AgI + e-}\]; E° = – 0.152 V
\[\ce{Ag -> Ag+ + e-}\]; E° = – 0.800 V
The value of log Ksp for AgI is ______.
On which electrode the oxidation reaction takes place?
Which of the following is incorrect?
Galvanic cell is a device in which ______.
Calculate the λ0m for Cl- ion from the data given below:
∧0m MgCl2 = 258.6 Scm2 mol-1 and λ0m Mg2+ = 106 Scm2 mol-1
What is an electrochemical cell? What does it consist of?
