Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction:
C6H5COCH3, CH3-CHO, CH3COCH3
Advertisements
उत्तर
Aldehydes are generally more reactive than ketones in nucleophilic addition reactions because of steric and electronic reasons. In case of ketones, aromatic ketones are more susceptible to nucleophilic attack than aliphatic ketones.
CH3COCH3 < C6H5COCH3 < CH3CHO
APPEARS IN
संबंधित प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Schiff’s base
Give plausible explanation for the following:
Cyclohexanone forms cyanohydrin in good yield but 2, 2, 6 trimethylcyclohexanone does not.
Alkenes 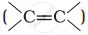 and carbonyl compounds
and carbonyl compounds 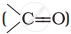 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Acetaldehyde and acetone differ in their reaction with
The most stable reagent for the conversion of R – CH2OH → RCHO is
What happens when propanone is treated with CH3MgBr and then hydrolysed?
What happens when ethanal is treated with excess ethanol and acid?
The product of following reaction is
\[\ce{CH3 - CH = CH - CH2 - CHO ->[i) LiAlH4][ii) H3O+]}\] ______?
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Give an example of the reaction in the following case.
Oxime
