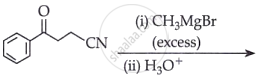Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction:
C6H5COCH3, CH3-CHO, CH3COCH3
Advertisements
उत्तर
Aldehydes are generally more reactive than ketones in nucleophilic addition reactions because of steric and electronic reasons. In case of ketones, aromatic ketones are more susceptible to nucleophilic attack than aliphatic ketones.
CH3COCH3 < C6H5COCH3 < CH3CHO
APPEARS IN
संबंधित प्रश्न
How are the following compounds prepared?
benzaldehyde from benzene
Write the products formed when CH3CHO reacts with the following reagents : HCN
What is meant by the following term? Give an example of the reaction in the following case.
Acetal
What is meant by the following term? Give an example of the reaction in the following case.
Semicarbazone
Which one of the following gives only one monochloro derivative?
What happens when ethanal is treated with excess ethanol and acid?
Draw the structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
The major product of the following reaction is: