Advertisements
Advertisements
प्रश्न
Answer briefly.
What is a carrier wave?
Advertisements
उत्तर
The high-frequency waves on which the signals to be transmitted are superimposed are called carrier waves.
APPEARS IN
संबंधित प्रश्न
How are infrared waves produced?
Identify the part of the electromagnetic spectrum which is:
produced by bombarding a metal target by high speed electrons.
A radio can tune in to any station in the 7.5 MHz to 12 MHz band. What is the corresponding wavelength band?
Given below are some famous numbers associated with electromagnetic radiations in different contexts in physics. State the part of the electromagnetic spectrum to which each belongs.
(a) 21 cm (wavelength emitted by atomic hydrogen in interstellar space).
(b) 1057 MHz (frequency of radiation arising from two close energy levels in hydrogen; known as Lamb shift).
(c) 2.7 K [temperature associated with the isotropic radiation filling all space-thought to be a relic of the ‘big-bang’ origin of the universe].
(d) 5890 Å - 5896 Å [double lines of sodium]
(e) 14.4 keV [energy of a particular transition in 57Fe nucleus associated with a famous high resolution spectroscopic method (Mössbauer spectroscopy)].
Give a reason for the following:
Long-distance radio broadcasts use short-wave bands. Why?
A wave has wavelength 50 Å.
- Name the wave.
- State its speed in vacuum.
- State its one use.
Which radiation is used for satellite communication?
What is the range of the wavelength of the following electromagnetic waves?
(A) Radio waves.
In a Coolidge tube, electrons strike the target and stop inside it. Does the target get more and more negatively charged as time passes?
Can a hydrogen atom emit characteristic X-rays?
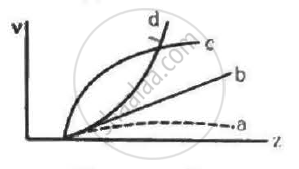
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
Find the energy, the frequency and the momentum of an X-ray photon of wavelength 0.10 nm.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Iron emits Kα X-ray of energy 6.4 keV. Calculate the times taken by an iron Kα photon to cross through a distance of 3 km.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Heat at the rate of 200 W is produced in an X-ray tube operating at 20 kV. Find the current in the circuit. Assume that only a small fraction of the kinetic energy of electrons is converted into X-rays.
The energy of a silver atom with a vacancy in K shell is 25.31 keV, in L shell is 3.56 keV and in M shell is 0.530 keV higher than the energy of the atom with no vacancy. Find the frequency of Kα, Kβ and Lα X-rays of silver.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
If the operating potential in an X-ray tube is increased by 1%, by what percentage does the cutoff wavelength decrease?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the scientist who discovered
X-rays
Name the scientist who discovered Microwaves
Name the part of the electromagnetic spectrum which is:
Produced by bombarding a metal target with high electrons.
Gamma rays and radio waves travel with the same velocity in free space. Distinguish between them in terms of their origin and the main application.
Name two sources of infrared radiation.
Solve the numerical problem.
The speed of light is 3 × 108 m/s. Calculate the frequency of red light of a wavelength of 6.5 × 10−7 m.
Name the e.m. waves which are suitable for radar systems used in aircraft navigation. Write the range of frequency of these waves.
The fundamental frequency of an open organ pipe is 300 Hz. The first overtone of this pipe has same frequency as first overtone of a closed organ pipe. If speed of sound is 330 m/s, then the length of closed organ pipe is:
The frequency of x-rays, y-rays and ultraviolet rays are respectively a, b and c. Then:-
What is time period of the light for which the eye is most sensitive?
Identify the electromagnetic wave whose wavelength range is from about 10-12 m to about 10-8 m. Write one use of this.
Assertion (A): Ultraviolet radiations is scattered more as compared to the microwave radiations.
Reason (R): Wavelength of ultraviolet radiation is more than the wavelength of microwave radiation.
What is the wavelength range of electromagnetic radiation used in radio broadcast?
