Advertisements
Advertisements
प्रश्न
Account for the following :
Manganese shows maximum number of oxidation states in 3d series.
Advertisements
उत्तर
Mnexhibits all the oxidation states from (+ 2) to (+ 7)
`"_25Mn = [Ar]3d^s 4s^2`
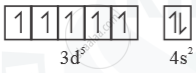
Mn+ =  Half filled (stable)
Half filled (stable)
Mn2+ = 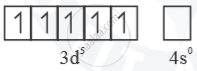
Mn3 = 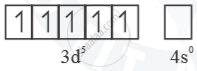
Mn4+= 
Mn5+ = 
Mn6+ = 
Mn+7 = 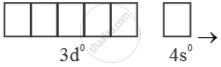 → Highest oxidation state = (+7)
→ Highest oxidation state = (+7)
APPEARS IN
संबंधित प्रश्न
Complete the following equations : 2 Na2CrO4 + 2 H + →
Complete the following equation : MnO4- + 8H+ + 5e- →
Complete the following equation :
Potassium permanganate on heating at 513 K gives a product which is ______.
Indicate the step in the preparation of K2Cr2O7 from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with oxalic acid? Write the ionic equation for the reaction.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
