Advertisements
Advertisements
प्रश्न
Account for the following :
Manganese shows maximum number of oxidation states in 3d series.
Advertisements
उत्तर
Mnexhibits all the oxidation states from (+ 2) to (+ 7)
`"_25Mn = [Ar]3d^s 4s^2`
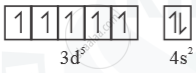
Mn+ =  Half filled (stable)
Half filled (stable)
Mn2+ = 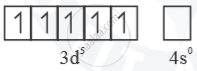
Mn3 = 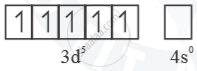
Mn4+= 
Mn5+ = 
Mn6+ = 
Mn+7 = 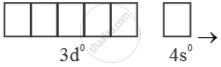 → Highest oxidation state = (+7)
→ Highest oxidation state = (+7)
APPEARS IN
संबंधित प्रश्न
Complete the following equation : MnO4- + 8H+ + 5e- →
Name the oxometal anions of the first series of the transition metals in which the metal exhibits the oxidation state equal to its group number.
Give an example and suggest a reason for the following feature of the transition metal chemistry:
The highest oxidation state is exhibited in oxoanions of a metal.
Indicate the steps in the preparation of K2Cr2O7 from chromite ore.
Answer the following question.
When MnO2 is fused with KOH in the presence of KNO3 as an oxidizing agent, it gives a dark green compound (A). Compound (A) disproportionates in an acidic solution to give a purple compound (B). An alkaline solution of compound (B) oxidizes KI to compound (C) whereas an acidified solution of compound (B) oxidizes KI to (D). Identify (A), (B), (C), and (D).
When \[\ce{Cu^2+}\] ion is treated with \[\ce{KI}\], a white precipitate is formed. Explain the reaction with the help of chemical equation.
KMnO4 is coloured due to ______.
The oxidation state of manganese in the product obtained in a reaction of potassium permanganate and hydrogen peroxide in a basic medium is ______.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
