Advertisements
Advertisements
Question
Account for the following :
Manganese shows maximum number of oxidation states in 3d series.
Advertisements
Solution
Mnexhibits all the oxidation states from (+ 2) to (+ 7)
`"_25Mn = [Ar]3d^s 4s^2`
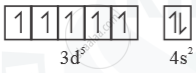
Mn+ =  Half filled (stable)
Half filled (stable)
Mn2+ = 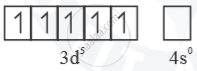
Mn3 = 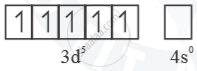
Mn4+= 
Mn5+ = 
Mn6+ = 
Mn+7 = 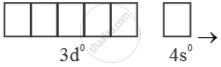 → Highest oxidation state = (+7)
→ Highest oxidation state = (+7)
APPEARS IN
RELATED QUESTIONS
Complete the following chemical equation
8MnO4- + 3S2O32- + H2O →
Complete the following equation :
`2MnO_4^(-)+6H^++5NO_2^(-)rarr`
Complete the following chemical equation
Cr2O72- + 3Sn2+ + 14H+ →
Name the oxometal anions of the first series of the transition metals in which the metal exhibits the oxidation state equal to its group number.
Describe the preparation of potassium dichromate from iron chromite ore. What is the effect of increasing pH on a solution of potassium dichromate?
Using IUPAC norms, write the formula of potassium trioxalatochromate(III).
\[\ce{KMnO4}\] acts as an oxidising agent in alkaline medium. When alkaline \[\ce{KMnO4}\] is treated with \[\ce{KI}\], iodide ion is oxidised to ______.
Complete the reaction mentioning all the products formed:
\[\ce{2KMnO4 ->[\Delta]}\]
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
