Advertisements
Advertisements
प्रश्न
A gas is taken through a cyclic process ABCA as shown in figure. If 2.4 cal of heat is given in the process, what is the value of J ?

Advertisements
उत्तर
Heat given in the process, ∆Q = 2.4 cal
∆W = WAB + WBC + WCA
For line AB:-
Change in volume,
ΔV = 0
∴ WAB = 0
For line BC :-
Mean pressure, \[P = \frac{1}{2} \times (100 + 200) kPa = 150 \times {10}^3 Pa\]
\[ ∆ V = (700 - 500) cc = 200 cc\]
\[ W_{BC} = 150 \times {10}^3 \times 200 \times {10}^{- 6} \]
\[ W_{BC} = 30 J\]
For line AC :-
P = 100 kPa
ΔV = 200 cc
WCA = 100 × 103 × 200 × 10−6
Total work done in the one cycle is given by
∆W = 0 + 150× 103 × 200 × 10−6 − 100 × 103 × 200 × 10−6
∆W = `1/2` × 300 × 103 × 200 × 10−6 − 20
∆W = 30 J − 20 J = 10 J
∆U = 0 .............(in a cyclic process)
∆Q = ∆U + ∆W
⇒ 2.4J = 10
\[\Rightarrow J = \frac{10}{2 . 4} = \frac{100}{24} = \frac{25}{6} = 4 . 17 \text{J/cal}\]
APPEARS IN
संबंधित प्रश्न
In changing the state of a gas adiabatically from an equilibrium state A to another equilibrium state B, an amount of work equal to 22.3 J is done on the system. If the gas is taken from state A to B via a process in which the net heat absorbed by the system is 9.35 cal, how much is the net work done by the system in the latter case? (Take 1 cal = 4.19 J)
Should the internal energy of a system necessarily increase if heat is added to it?
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
An ideal gas is pumped into a rigid container having diathermic walls so that the temperature remains constant. In a certain time interval, the pressure in the container is doubled. Is the internal energy of the contents of the container also doubled in the interval ?
When a tyre bursts, the air coming out is cooler than the surrounding air. Explain.
Consider the process on a system shown in figure. During the process, the work done by the system ______________ .
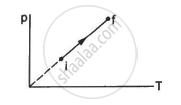
An ideal gas goes from the state i to the state f as shown in figure. The work done by the gas during the process ______________ .
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
One gram of water (1 cm3) becomes 1671 cm3 of steam at a pressure of 1 atm. The latent heat of vaporization at this pressure is 2256 J/g. Calculate the external work and the increase in internal energy.
derive the relation between the change in internal energy (∆U), work is done (W), and heat (Q).
The internal energy of a system is ______
When 1 g of water at 0° C and 1 x 105 N/m2 pressure is converted into ice of volume 1.082 cm3, the external work done will be ____________.
In insulated systems, the amount of external work done by the gas is proportional to:
Figure shows the P-V diagram of an ideal gas undergoing a change of state from A to B. Four different parts I, II, III and IV as shown in the figure may lead to the same change of state.

- Change in internal energy is same in IV and III cases, but not in I and II.
- Change in internal energy is same in all the four cases.
- Work done is maximum in case I
- Work done is minimum in case II.
An expansion process on a diatomic ideal gas (Cv = 5/2 R), has a linear path between the initial and final coordinates on a pV diagram. The coordinates of the initial state are: the pressure is 300 kPa, the volume is 0.08 m3 and the temperature is 390 K. The final pressure is 90 kPa and the final temperature s 320 K. The change in the internal energy of the gas, in SI units, is closest to:
The molar specific heat of He at constant volume is 12.47 J/mol.K. Two moles of He are heated at constant pressure. So the rise in temperature is 10 K. Find the increase in internal energy of the gas.
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
